Optical Dendrometry reliably captures water potential in branches during lab drying
Abstract
Measuring water potential is critical to any investigation into plant hydraulics and water deficit response. However, methodological constraints to continuously monitoring water potential endure as a key hurdle in many experimental designs. Optical Dendrometry provides key utility in monitoring plant water status through continuous image capture, mapping tissue diameter to water potential change. However, the technique has thus far been rigorously limited to fully expanded tissues (petioles, lamina, coniferous branchlets) to avoid the confounding effect of tissue secondary growth. Alternatively, here we test the application of Optical Dendrometry to growing branchlets during water deficit to examine the relationship between water potential and branch diameter in intact plants under drought-to-death conditions. We investigate the premise that branch diameter and water potential should become linearly correlated once xylem water potentials decline beyond thresholds of cell-expansion. Under experimental drought from fully hydrated to complete mortality, Eucalyptus crebra branch diameter and water potential were highly correlated (R2 = 0.99), highlighting a consistent linear relationship between hydration status and branch diameter once the growth signal is eliminated. The finding provides a useful option for continuous water potential monitoring in drying plants where non-expanding tissues are logistically unavailable. This potentially simplifies hydraulic experiments by removing tissue selection constraints, allowing Optical Dendrometry under drought-to-death experimental scenarios to be applied to any convenient small branch.
Introduction
As the field of plant hydraulics continues to rapidly grow in scope, we have an ever-increasing need for an accessible, non-destructive means of continuously monitoring plant water status at high temporal resolution and precision. Achieving accurate water potential measurement is essential for understanding the timing and onset of plant water stress, loss of productivity and ultimately tissue death (Brodribb, Powers, Cochard, & Choat, 2020; Choat et al., 2012). Thermocouple psychrometry (Tyree & Sperry, 1989) remains highly temperature-dependent, and pressure chamber (Scholander, Bradstreet, Hemmingsen, & Hammel, 1965) measurements are inherently destructive and non-continuous by nature. Instruments such as Florapulse micro-tensiometers provide long-term continuous measurements of water potential through stem-embedded sensors (Lakso, Santiago, & Stroock, 2022). The Optical Dendrometry approach (Bourbia & Brodribb, 2023; Bourbia, Lucani, & Brodribb, 2022; Bourbia, Lucani, Carins‐Murphy, Gracie, & Brodribb, 2023) instead utilises non-destructive continuous image capture to assess tissue diameter change against hydration status, mapping a focal tissue area change against Scholander pressure chamber measurements to produce sub-minute monitoring of water potential (commercial: cavicams.com, or open-source: opensourceov.org). Echoing this method, Contact Dendrometers also measure tissue diameter change over water potential (Gleason et al., 2024). A defining characteristic of the Optical Dendrometry method is that the cameras must be placed on lamina, petioles or branchlets (Haverroth, Da-Silva, Taggart, Oliveira, & Cardoso, 2024; Manandhar, Pichaco, & McAdam, 2024; Manandhar, Rimer, et al., 2024), specifically in tissues exhibiting no or limited secondary growth, as these mature tissues reveal linear relationships between tissue shrinkage and water potential. This careful placement avoids the complications that arise with examining growing tissues under fluctuating water potentials (Bourbia & Brodribb, 2023; Gleason et al., 2024), excluding potentially confounding growth signal of the plants.
By contrast, in growing tissues, traditional dendrometry is a well-established method for capturing fluctuations in plant diameter relating to environmental variables, particularly in woody forest trees (Sevanto, 2025; Steppe, De Pauw, Lemeur, & Vanrolleghem, 2006). Most recently, concerted effort has been dedicated towards disentangling the relationship between tissue growth during periods of water abundance and shrinkage during water stress (Peters et al., 2025; Sevanto, 2025; Ziegler, Grote, Alongi, Knüver, & Ruehr, 2024; Zweifel, Haeni, Buchmann, & Eugster, 2016); partitioning cycles of cell expansion and shrinkage with the goal of monitoring tree water deficit (TWD) on diurnal and seasonal time scales. The challenges in field dendrometry modelling arise due to the complexity of segmenting cambium cell division and hydration state during cycles of drought and rehydration (Peters et al., 2025; Steppe et al., 2006). Despite the complexities involved in partitioning growth and water potential signals, these studies provide valuable insight into the amplitude of hydraulic stress in forests (Peters et al., 2025; Sevanto, 2025), suggesting a close correlation between stem diameter and water potential during drying.
Here we investigate the application of the Optical Dendrometry method in growing branches to examine dendrometry under lab-imposed drought. We hypothesise that the branch Dendrometry signal should become linearly correlated with xylem water potential once plants begin to dehydrate beyond their tissue diameter maxima (the point at which growth ceases and tissues begin to shrink under water stress), effectively eliminating the growth signal in drying plants. Using Eucalyptus crebra, an arid-adapted eucalypt species (Bourne, Creek, Peters, Ellsworth, & Choat, 2017), we apply Optical Dendrometry using “Dendrocams” (cavicams.com) on branches ~5 mm diameter with paired stem psychrometry with the aim of determining the precise relationship between branch tissue shrinkage and water potential during terminal drying.
Materials and Methods
Plant material
Eucalyptus crebra trees were grown at Western Sydney University Hawkesbury Institute for the Environment (Richmond, NSW, Australia). Trees were grown in 10 L pots into potting mix in filtered natural light with daily drip irrigation until ~1.5 m in height, with stems approx. 8 mm diameter. Four plants of uniform developmental age were used to compare the relationship between water potential and branch diameter during experimental drying under lab conditions. Paired ICT stem psychrometers (Armadale, NSW, Australia) and Optical Dendrometers (Hobart, Tasmania, Australia) were installed on each plant capturing the continuous dry-down.
Measurement of water potential and Optical Dendrometry
Plants were transferred to the laboratory for experimental drying under controlled conditions at 20-25oC under stable low light conditions of ~10 µmol quanta m-2s-1. Pots were removed to expose root-soil volume to air, increasing the rate of drying but leaving soil volumes largely intact. An ICT stem psychrometer was applied to a central region of stem close to the first branches, approx. 30 cm from the root collar. A region of epidermis was removed with a razor blade to expose the xylem and was washed with distilled water. The psychrometer chamber was clamped onto the stem and sealed with layers of parafilm before clamping. Plants took 8-14 days to dry to xylem water potentials below -7 MPa, beyond the known species P88 of -6.41 MPa (Bourne et al., 2017).
On the same plants, a subtending branch or terminal segment of stem approx. 5mm in diameter supporting living leaves was selected and a Dendrocam (cavicam.co) Optical Dendrometry monitoring camera (Bourbia & Brodribb, 2023; Bourbia et al., 2023) was attached to capture changes in tissue diameter as the plant dried and was set to log in five-minute intervals until the completion of plant drying (Fig. S1).
Statistical analysis
We used a linear regression to examine the relationship between Optical Dendrometry signal and the rate of water potential decline measured using psychrometry, determining success of fit using R2. To ensure a linear relationship, we compared this linear model to a sigmoidal nonlinear model using AIC, and summarised model performance with ΔAIC. All analysis was performed using R (R, 2024) version 4.5.1.
Results
In all plants, the Optical Dendrometry signal on small branches was closely aligned with stem psychrometer signal once plants began to dry (Fig. 1A). All plants showed a highly correlated linear relationship between water potential and branch Optical Dendrometry output, with a mean R2 of 0.99, and a minimum R2 of 0.984 (Fig. 1B). Comparison with a non-linear sigmoidal model showed no increase in predictive power compared with the linear model (ΔAIC = 1.34), supporting the highly consistent linear relationship between branch diameter and water potential. The Optical Dendrometry signal revealed greater resolution in data capture with less noise (measurement error) than psychrometry, particularly in cases of temperature fluctuation (Fig. S2).
A) Dehydration of Eucalyptus crebra small trees (~1.5 m) under controlled lab conditions showing continuous Optical Dendrometry (black line) in branch (~5 mm diameter) area over elapsed hours, with time 0 set at maximum hydrated branch area, scaled from the maximum as 100%. Blue lines (second axis) show corresponding psychrometer measurement of stem water potential per plant. B) Linear regressions (red line) between branch area and water potential displaying R2 correlation per plant.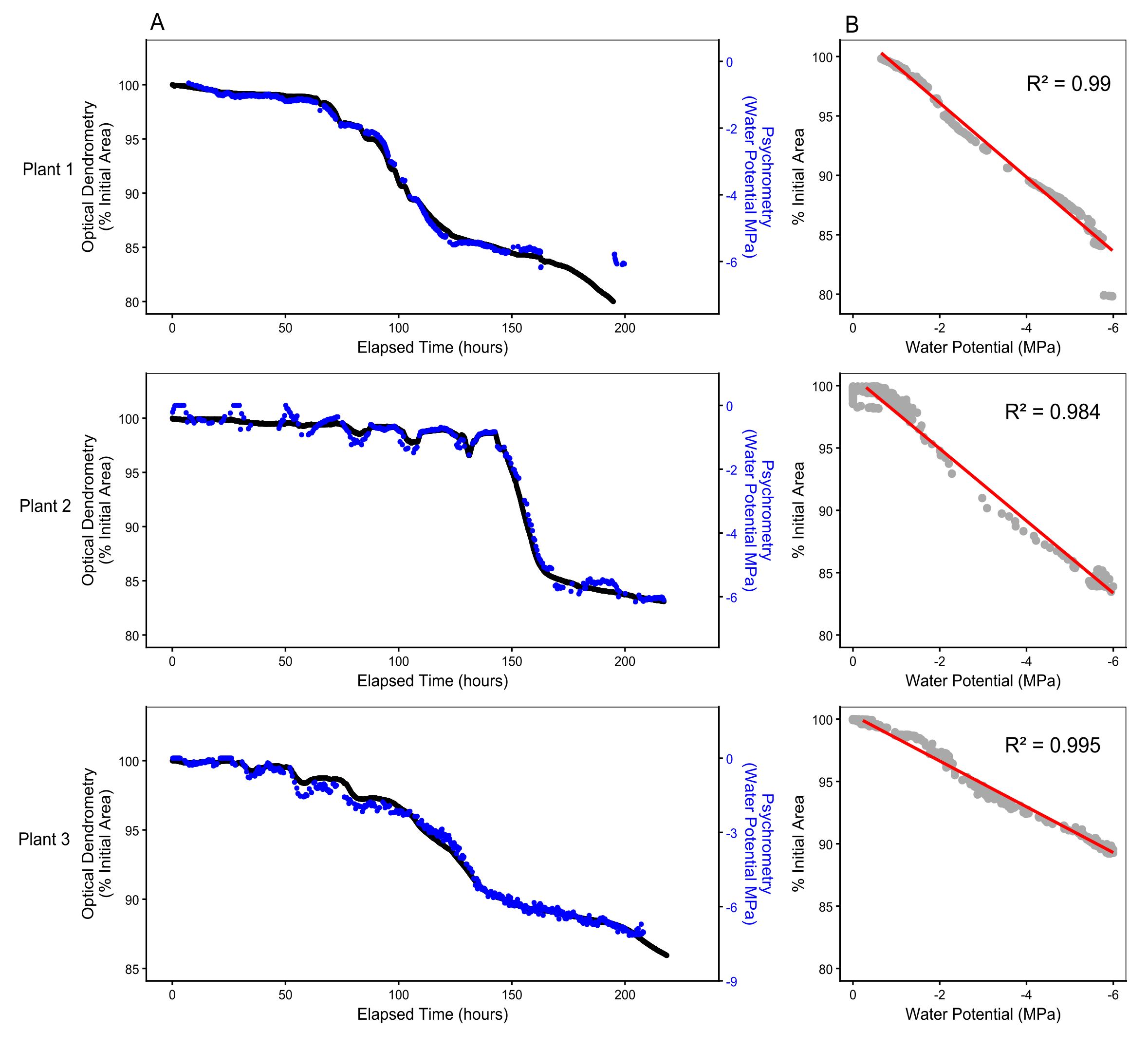
Figure 1: Optical Dendrometry signal and stem Psychrometer water potential measurement correlate closely in branches of three plants over the dehydration gradient.
Discussion
Here our results extend the methodological foundations established by previous Optical Dendrometry studies by revealing that small branches of an example eucalypt species with rapid secondary growth potential exhibit a highly consistent linear relationship (R2 of 0.99) between tissue diameter and xylem water potential under drought-to-death conditions. This finding opens an additional avenue for monitoring plant water potential by expanding available tissues for Optical Dendrometry placement. By working within a drought-to-death context, we demonstrate that conveniently accessible small branches can serve as a reliable proxy for water potential, enhancing the practicality of drought monitoring.
Our experiment highlights that when tissues shrink below the maximum hydrated tissue diameter, growth-related signals become subsumed by the strong water potential signal. Once branch radial growth slows or ceases during drying, tissue diameter becomes governed largely by plant water status, thereby fulfilling the core assumption of the Zero-Growth model (Steppe et al., 2006; Zweifel et al., 2016) (Fig. S2). This aligns with Zweifel et al. (2016), who demonstrated that drought-driven radial shrinkage is largely attributable to reversible water loss rather than cambial activity. This drought-driven simplification of radial signals reinforces emerging tree water-balance models (Peters et al., 2025) which aim to algorithmically separate reversible water-deficit signals from growth-related components embedded within stem diameter records (Özçelik, 2025). Our results indicate that, under lethal drought, the environment itself acts as this “signal separator,” making water deficit the only driver of diameter variation and enabling direct linear calibration without complex computation. Ziegler et al. (2024) argues that under severe drought, radial variation becomes nearly a one-to-one proxy for xylem water potential because the loss of turgor eliminates growth-driven morphological changes. Our observations provide direct empirical support for this prediction.
These data suggest that Optical Dendrometry can be applied on branches with the same effectiveness as non-expanding tissues in a lab setting, offering paired use with Optical Vulnerability for the precise quantification of embolism resistance. Given that “Dendrocams” (cavicam.co) are easy to install, non-destructive, and measure in high-frequency, this form of branch Dendrometry offers potential improvement over traditional thermocouple psychrometry, particularly given that psychrometers remain open to artefact even in highly controlled lab-based experiments (Fig. S3). Concurrent pressure-chamber measurements are still required to calibrate this signal to water potential, and the limited field of view (< 1 cm2) of current Dendrocam hardware constrains the branch sizes that can be observed, although this restriction could be alleviated by future hardware improvements.
As droughts continue to intensify, we provide a simplified method for assessing plant water status, showing that branch dendrometry should reflect a clear and calibratable water potential signal during terminal drought in this lab-based scenario. Branch Optical Dendrometry may have applied lab-monitoring applications in plants that cannot support non-growing tissue Optical Dendrometry (i.e. short petioles or leaf death in cameras), potentially reducing the logistical burden of tissue selection and increasing options for coverage. However, this experiment was conducted on a single eucalypt species, and further testing is needed before these findings can be generalized across species. While the practical application of this method is within a lab-setting to expand placement options of Optical Dendrometers, the conceptual implications of the linear relationship could theoretically help parameterise traditional field-based dendrometry models in acute drought scenarios (Bourbia, Yates, & Brodribb, 2025), helping to disentangle overlapping growth and hydraulic processes (Sevanto, 2025; Ziegler et al., 2024).
Acknowledgements
Thanks to the technical team at Western Sydney University Hawkesbury Institute for the Environment for help in procuring and maintaining plant material and lab equipment.
Author contributions
BC, BLHD and CY designed the conceptual framework. BLHD, CY, YZ and RH performed the laboratory experiments. BHLD performed the analysis and interpretation of data in consultation with BC. BLHD wrote the manuscript with contributions from all authors.
References
- Bourbia, I., & Brodribb, T. J. (2023). A new technique for monitoring plant transpiration under field conditions using leaf optical dendrometry. Agricultural and Forest Meteorology, 331, 109328. https://doi.org/10.1016/j.agrformet.2023.109328
- Bourbia, I., Lucani, C., & Brodribb, T. J. (2022). Constant hydraulic supply enables optical monitoring of transpiration in a grass, a herb, and a conifer. Journal Of Experimental Botany, 73(16), 5625-5633. https://doi.org/10.1093/jxb/erac241
- Bourbia, I., Lucani, C., Carins‐Murphy, M. R., Gracie, A., & Brodribb, T. J. (2023). In situ characterisation of whole‐plant stomatal responses to VPD using leaf optical dendrometry. Plant, Cell & Environment, 46(11), 3273-3286. https://doi.org/10.1111/pce.14658
- Bourbia, I., Yates, L. A., & Brodribb, T. J. (2025). Using long‐term field data to quantify water potential regulation in response to VPD and soil moisture in a conifer tree. New Phytologist, 246(3), 911-923. https://doi.org/10.1111/nph.70056
- Bourne, A. E., Creek, D., Peters, J. M. R., Ellsworth, D. S., & Choat, B. (2017). Species climate range influences hydraulic and stomatal traits in Eucalyptus species. Annals of Botany, 120(1), 123-133. https://doi.org/10.1093/aob/mcx020
- Brodribb, T. J., Powers, J., Cochard, H., & Choat, B. (2020). Hanging by a thread? Forests and drought. Science, 368(6488), 261-266. https://doi.org/10.1126/science.aat7631
- Choat, B., Jansen, S., Brodribb, T. J., Cochard, H., Delzon, S., Bhaskar, R., . . . Zanne, A. E. (2012). Global convergence in the vulnerability of forests to drought. Nature, 491(7426), 752-755. https://doi.org/10.1038/nature11688
- Gleason, S. M., Stewart, J. J., Allen, B., Polutchko, S. K., McMahon, J., Spitzer, D., & Barnard, D. M. (2024). Development and application of an inexpensive open-source dendrometer for detecting xylem water potential and radial stem growth at high spatial and temporal resolution. AoB PLANTS, 16(2). https://doi.org/10.1093/aobpla/plae009
- Haverroth, E. J., Da-Silva, C. J., Taggart, M., Oliveira, L. A., & Cardoso, A. A. (2024). Shoot hydraulic impairments induced by root waterlogging: Parallels and contrasts with drought. Plant Physiology, 197(1). https://doi.org/10.1093/plphys/kiae336
- Lakso, A. N., Santiago, M., & Stroock, A. D. (2022). Monitoring stem water potential with an embedded microtensiometer to inform irrigation scheduling in fruit crops. Horticulturae, 8(12), 1207. https://doi.org/10.3390/horticulturae8121207
- Manandhar, A., Pichaco, J., & McAdam, S. A. M. (2024). Abscisic acid increase correlates with the soil water threshold of transpiration decline during drought. Plant, Cell & Environment, 47(12), 5067-5075. https://doi.org/10.1111/pce.15087
- Manandhar, A., Rimer, I. M., Soares Pereira, T., Pichaco, J., Rockwell, F. E., & McAdam, S. A. M. (2024). Dynamic soil hydraulic resistance regulates stomata. New Phytologist, 244(1), 147-158. https://doi.org/10.1111/nph.20020
- Özçelik, M. S. (2025). Dendrometer-Based Analysis of Intra-Annual Growth and Water Status in Two Pine Species in a Mediterranean Forest Stand Under a Semi-Arid Climate. Forests, 16(8), 1229. https://doi.org/10.3390/f16081229
- Peters, R. L., Basler, D., Zweifel, R., Steger, D. N., Zhorzel, T., Zahnd, C., . . . Kahmen, A. (2025). Normalized tree water deficit: an automated dendrometer signal to quantify drought stress in trees. New Phytologist, 247(3), 1186-1198. https://doi.org/10.1111/nph.70266
- Scholander, P. F., Bradstreet, E. D., Hemmingsen, E., & Hammel, H. (1965). Sap Pressure in Vascular Plants: Negative hydrostatic pressure can be measured in plants. Science, 148(3668), 339-346. https://doi.org/10.1126/science.148.3668.339
- Sevanto, S. (2025). Dendrometers-what are they good for? Tree Physiology, 45(4). https://doi.org/10.1093/treephys/tpaf035
- Steppe, K., De Pauw, D. J. W., Lemeur, R., & Vanrolleghem, P. A. (2006). A mathematical model linking tree sap flow dynamics to daily stem diameter fluctuations and radial stem growth. Tree Physiology, 26(3), 257-273. https://doi.org/10.1093/treephys/26.3.257
- Tyree, M. T., & Sperry, J. S. (1989). Vulnerability of xylem to cavitation and embolism. Annual Review of Plant Physiology and Plant Molecular Biology, 40(1), 19-36. https://doi.org/10.1146/annurev.pp.40.060189.000315
- Ziegler, Y., Grote, R., Alongi, F., Knüver, T., & Ruehr, N. K. (2024). Capturing drought stress signals: the potential of dendrometers for monitoring tree water status. Tree Physiology, 44(12). https://doi.org/10.1093/treephys/tpae140
- Zweifel, R., Haeni, M., Buchmann, N., & Eugster, W. (2016). Are trees able to grow in periods of stem shrinkage? New Phytologist, 211(3), 839-849. https://doi.org/10.1111/nph.13995
Attachments
Other
DownloadArticle statistics
 Views: 599
Views: 599
Downloads
 XML: 46
XML: 46
 PDF: 143
PDF: 143
