Can micro-tensiometers move us beyond the Scholander pressure chamber?
Abstract
Measurements of water potential are fundamental to the field of plant water relations, but we still rely almost exclusively on a single instrument, developed in mid-20th century, for its measure. More recently new technologies such as micro-tensiometers have been developed, providing an automated means to measure water potential in near real-time. What is the potential for these devices to move us beyond the Scholander pressure chamber?
The gold standard
The gold standard for measuring plant water status has remained the same for the past 70 years: the pressure chamber (i.e., the pressure bomb). This observation is remarkable. It’s striking that despite the rapid technological development over the past 70 years, we still rely on the same instrument developed in the late 1950s and early 1960s by Swedish-born Per Fredrik Scholander and his team at the Scripps Institution of Oceanography (University of California, San Diego).
The original development of the pressure chamber was squarely focused on a single goal, the proof of the cohesion-tension theory of water movement in plants (Scholander et al., 1965). This theory seems to have an enduring attraction for controversy, which has reoccurred since its inception. However, each time a study emerged that challenged the cohesion-tension theory (e.g., Canny, 1997; Zimmermann et al., 1994) it has invariably been shown to be artifactual upon closer examination (e.g., Cochard et al., 2000, 2001; Torres-Ruiz et al., 2015; Wei et al., 1999).
Over time, the pressure chamber became arguably the most critical instrument in advancing the field of plant water relations. Prior to its invention, the only direct means of measuring water potential was via thermocouple psychrometers. These measurements are extremely time consuming, confined to the lab, and technically demanding due to their strict thermostability requirements (Richards & Ogata, 1958; Spanner, 1951). The pressure chamber provided a relatively easy means by which scientists could quantitatively delimit phenomena in relation to the highly dynamic “tension” within the cohesion-tension framework. This is evidenced by the ubiquitous use of water potential (Ψ) as the independent variable in studies revealing hydraulic function across a huge range of species and contexts. Although the pressure chamber is relatively heavy and requires an associated gas tank, it remains relatively portable, allowing transport over long distances in the field. Or alternatively, excised leaves of certain species can be stored and transported for multiple days with no loss in accuracy with proper protocols (Rodriguez-Dominguez et al., 2022). From a practical point of view, the pressure chamber has proven its utility in the context of agriculture as a means to assess plant water status to guide irrigation management. This is especially true for perennial fruit crops, for which many have well-developed Ψ thresholds (e.g., Mirás-Avalos & Araujo, 2021; Moriana et al., 2012).
However, the pressure chamber has limits as well. The pressure chamber requires a manual, destructive measurement and is time-consuming (relative to the speeds at which water status can potentially change). Thus, it remains impractical for experimental designs requiring high throughput, or high spatial and/or temporal resolution. In the context of agriculture, one simply cannot do enough measurements to be accurate across large spatial scales, and thus its efficacy is reduced to providing “spot checks”. Finally, although the pressure chamber remains the gold standard for accuracy, extreme drought can induce leaf tissue damage undermining measurement reliability (Rodriguez-Dominguez et al., 2025).
Maybe these limits should come as no surprise given that the pressure chamber was developed to provide direct measurements in support of the cohesion-tension theory and not as an applied tool. In the original Scholander et al. (1965), there is no speculation of any practical application for the instrument regarding agriculture. Only later over time did the value and versatility of using the pressure chamber to measure Ψ gain more importance, eventually becoming the cornerstone of our understanding of plant water relations and an instrument widely used in agriculture for determining key crop water stress thresholds and guiding irrigation management (e.g., from Boyer, 1967; Grimes & Yamada, 1982 onward).
A new generation of devices
The value of automated, real-time measurements of plant water status has become increasingly evident over the years. This has driven the development of numerous approaches that attempt to measure Ψ via proxies. Examples include using dendrometers (traditional; Ziegler et al., 2024 and the more recent optical; Bourbia & Brodribb, 2023), leaf and/or canopy temperature (Cohen et al., 2005), spectroscopy (Cozzolino, 2017; De Bei et al., 2011), leaf turgor pressure (Zimmermann et al., 2008), and more. The sheer number and scope of attempted technologies underscores the value of the measurement, while the reality that the vast majority of these technologies have never been widely adopted highlights the difficulty of the challenge.
More recently, micro-tensiometers have been developed that measure plant tension directly (Pagay et al., 2014; and see subsequent works in Supplemental File 1). These are essentially miniaturized, diaphragm-based tensiometers that use a microelectromechanical (MEMS) pressure transducer coupled through a nanoporous membrane. Their measurement range is larger than traditional tensiometers with a theoretical limit of –10 MPa (Pagay et al., 2014) although practical limits appear to be much less negative, currently around -4 MPa (Haberstroh et al., 2025; and see Figure 1 below). The most recent micro-tensiometer versions are quite small (from 1 cm in diameter down to 3mm) allowing for installation into the trunk xylem of relatively small trunks/branches.
Once installed micro-tensiometer measurements are automated and thus can reveal the dynamics of changing Ψ in a manner that comes much closer to (but is not) real-time. Because plant hydraulics can change on very short timescales (e.g., microseconds during embolism formation) I will use the term “near” real-time to differentiate. Micro-tensiometer measurements typically require about 15-20 minutes for equilibration, which limits the measurement interval, although some studies report faster response times (Villalobos et al., 2025). This is certainly not fast enough to capture extremely fast events and may be only marginally effective in capturing phenomena occurring over similar time frames such as stomatal oscillations (Dzikiti et al., 2007; Prytz et al., 2003; Wallach et al., 2010).
I do not think it is hyperbole to suggest that automated, near real-time Ψ monitoring could open whole new fields of study. Examples could include the study of: 1) nighttime phenomena, which are underexplored for the simple reason that humans prefer to sleep at night, or 2) ecological systems that are physically inaccessible, or 3) rare and/or unpredictable events which are extremely difficult to capture such as responses to extreme weather events; and there are surely others.
However, micro-tensiometers are not without their disadvantages. Although current micro-tensiometers (i.e., FloraPulse; https://florapulse.com) are reasonably priced their cost is still high enough to preclude them from being deployed at a very high density, or frequently across large spatial scales. For comparison consider temperature sensors that can be almost 1/100 the cost. In addition, the instruments are not nearly as durable as a pressure chamber and require frequent replacing. Installing the micro-tensiometers requires drilling a hole in the trunk, which can provoke a wounding response in some species that can render the probes inoperable or decrease their measurement lifespan significantly (Lakso et al., 2022). In other cases the installation could result in trunk cracking inducing cavitation (Di Biase et al., 2025).
Micro-tensiometer accuracy
The advantages and disadvantages of micro-tensiometers are outlined above, but how accurate are they? To date, numerous studies have attempted to validate micro-tensiometer measurements with paired pressure chamber measurements (See Supplemental File 1). Here, the data presented in those works was comprehensively extracted using WebPlotDigitizer (https://automeris.io/wpd/) and the constructed database is shared in Supplemental File 2.
When viewed globally, micro-tensiometer measurements are quite variable when compared with their paired Ψleaf or Ψstem measurements using a pressure chamber (Fig. 1A). Generally, there is greater variability when micro-tensiometer values are compared to Ψleaf than to Ψstem. In addition, micro-tensiometer values tend to be much less negative when compared to Ψleaf. When relationships with Ψstem are examined in more detail (Fig. 1B) we see that for all species there is a strong correlation between the values, although the correlation does not always lie on the 1:1 line. Regression analyses by species are all highly significant, but the strength of those correlations varied, with r2 values ranging from 0.45 for grape to 0.97 for almond (Fig. 1B & Supplemental File 2). This contrasts with correlations with Ψleaf which can be quite low; for example with r2 of 0.21 and 0.38, reported by Pagay, 2022 for grape and Conesa et al., 2023 for nectarine, respectively.
We may expect micro-tensiometer values (measured in the trunk) to diverge from Ψleaf values because of their relative positions in the plant’s hydraulic pathway, and likewise for micro-tensiometer values to be more similar to Ψstem. Across works, the variability between measurements appears to arise from numerous factors that include the time of the measure (Blanco & Kalcsits, 2021, 2023; Conesa et al., 2023; Haberstroh et al., 2025; Pagay, 2022), relative height from which the pressure chamber measure was taken (Haberstroh et al., 2025; Lakso et al., 2022), and capacitance or “lag” effects where the water potential takes time to equilibrate across tissues following rapid changes in water supply or demand (Christenson et al., 2024; Lakso et al., 2022; Pagay, 2022). This substantial variability raises the question if micro-tensiometers are accurate enough for dissecting fine level physiology. Certainly their use necessitates cross-validation with a proven instrument like the pressure chamber.
Today we have a whole field established upon relating behaviors to pressure chamber measurements in leaves (e.g., predawn, stem, leaf). Yet, the meta-analysis above demonstrates that these relationships do not directly translate to trunk micro-tensiometer measurements. The significant correlations (notably with Ψstem) suggest that the parallels are strong. However, the values themselves will not be created equal due to upstream differences in hydraulic conductivity, local capacitance differences, sectoriality, and in some cases hydraulic vulnerability segmentation. Therefore, incorporating Ψtrunk via micro-tensiometers will require some significant adaptation and integration. Certainly leaves, being the key control point for the regulation of transpiration, will remain a critical organ for the study of plant hydraulics which trunk measurements simply cannot replace.
One remaining question is if micro-tensiometers accurately measure the Ψtrunk itself? The wounding response, and/or cavitation during installation, are potential sources of error highlighted in some works (Di Biase et al., 2025; Lakso et al., 2022). Alternatively, the installation location of the micro-tensiometer may not represent the predominant Ψtrunk. In other words, the assumption that Ψtrunk is perfectly equilibrated radially across the trunk may not hold. The most conspicuous evidence to this effect is the large differences in sap flow rates across the trunk radius in numerous species (e.g. Clearwater et al., 1999; Delzon et al., 2004; and many other works).
Questions remain regarding the accuracy of micro-tensiometers, but what are some examples of how this technology can be used?
A) Global overview of the paired measurements categorized by the type of pressure chamber measure that was used for comparison; either leaf (red) or stem (cyan) water potential. B) Comparison of only the micro-tensiometer and stem water potential measurements categorized by species. Results from independent regression analyses by species can be found in a dedicated tab in Supplemental File 2. 1:1 lines are shown as the dashed black lines.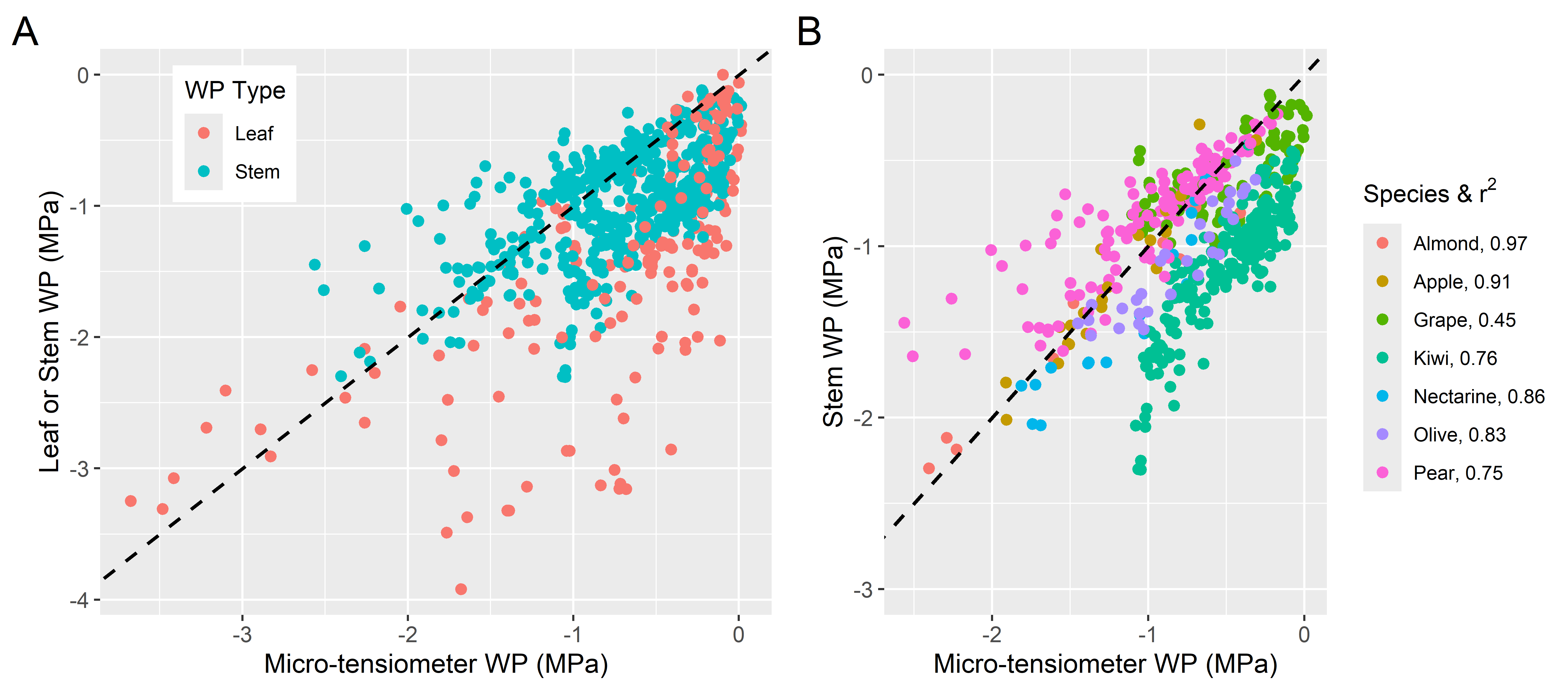
Figure 1: Meta-analysis of paired micro-tensiometer and pressure chamber measurements.
The steady state conundrum
Plant water relations, including Ψ, are highly dynamic across an enormous range of time scales, from less than a second, to minutes, to hours, to months, to seasons. There is a bit of irony in the fact that our understanding of plant water relations is founded almost entirely on steady-state scenarios. This persists because dynamic scenarios are extremely complex to approach experimentally, and while steady state scenarios are surely incomplete, they still soundly elucidate a ton of phenomena. With a dose of poetic license, this is analogous to the relationship between Newtonian and quantum physics: Newtonian physics cannot explain quantum behavior, but it remains perfectly functional for the scales on which Newton worked. Nature is the proof that contradictory phenomena reconcile across scales, regardless if we are capable of understanding how. Having technologies that allow near real-time Ψ measurements holds the potential to reveal, and then study, these more dynamic behaviors.
How low can we go
Many important plant hydraulic phenomena exhibit threshold behavior dependent on Ψ. This means that they are more or less stable until a critical Ψ is reached, at which point they undergo a rapid non-linear change. This is why whether studying ecosystems or agriculture, the most severe level of water stress experienced, or Ψmin, is an extremely important parameter. One can refer to a daily Ψmin, or even a seasonal Ψmin, but in any case this value represents the most negative Ψ during any defined period of time.
The seasonal and diurnal dynamics of Ψ leads to some inaccuracy in our ability to measure Ψmin with a pressure chamber. Scientists do their best at capturing this Ψmin, but because Ψ is constantly changing the will always be some error. Near real-time measurements can help decrease this uncertainty and allow for a more accurate determination of diurnal or seasonal Ψmin (assuming uninterrupted measures). This is not without caveats however. Based on the meta-analysis above micro-tensiometer Ψmin values may not correspond directly to Ψmin values in stems or leaves. Furthermore, there may be lags due to capacitance effects that result in differences between organs in the timing of Ψmin during the day. Thus, although this could be extremely important for making more accurate predictions about how certain species or even ecosystems will behave, or more reliably identifying irrigation thresholds in an agricultural setting, it requires reconciling trunk, stem, and leaf Ψmin.
Pressure and flux
Having near real-time Ψ measurements becomes more powerful when we consider pairing this data with measurements of flux. This potentially allows for studies aimed at revealing the dynamic regulation of numerous different hydraulic phenomena, including hydraulic conductivity.
One of the aspects of moving from a steady state to a dynamic scenario that is so daunting is the difficulty in disentangling contributions of hydraulic conductivity (soil, plant, in serial vs parallel, etc.). Some recent approaches that have tried to attack this challenge have been extremely fruitful, but caution is warranted since many do not incorporate the dynamics of conductivity in certain plant organs, especially the roots system (Caldeira et al., 2014; Henzler et al., 1999; Vandeleur et al., 2009). Having near real-time pressure flux relationships can help reveal these dynamics.
Near real-time pressure flux relationships also hold the potential for elucidating other aspects of plant water relations that have been particularly challenging. One example of this is whole plant hydraulic capacitance. Previously, quantifying whole plant capacitance has leveraged diurnal pressure flux relationships using sap flow and psychrometers for example (e.g. Meinzer et al., 2003) or optical dendrometers (e.g. Bourbia & Brodribb, 2023). Having paired weighing lysimeter and micro-tensiometer measurements makes the type of approach presented in Meinzer et al. (2003) arguably more feasible, in that it allows the disentangling of transpiration and storage fluxes.
Figure 2 shows one day’s pressure flux relationship from a mini-lysimeter experiment in grapevine of a large mature vine in a 44 liter pot equipped with a micro-tensiometer in the trunk (data from Dell’acqua et al., 2025). The pressure (∆P, or the difference between the maximum predawn Ψtrunk and the Ψtrunk for each hour), flux (E) relationship during the daytime hours is quite linear, but breaks down during the nighttime hours, presumably because of capacitance refilling. If we assume the soil-trunk hydraulic conductance from 9-16h (9.7 mmoles m-2 s-1 MPa-1) remains stable then we can use this conductance to calculate the corresponding theoretical flux during the evening hours. This theoretical flux will be the sum of the water being transpired through the plant (which is measured by the lysimeter), but also the unknown volume of water refilling the capacitance of the vine. Relating this volume to the nighttime pressure drop gives you the plant capacitance (assuming sensor capacitance is negligible).
These are just a few examples, but clearly the power of these pressure flux relationships has the potential to open new realms for the plant hydraulic community.
Data shown was from measured from a grapevine on a mini-lysimeter equipped with a micro-tensiometer (data from Dell’acqua et al., 2025). Each point is labeled by the hour of measure.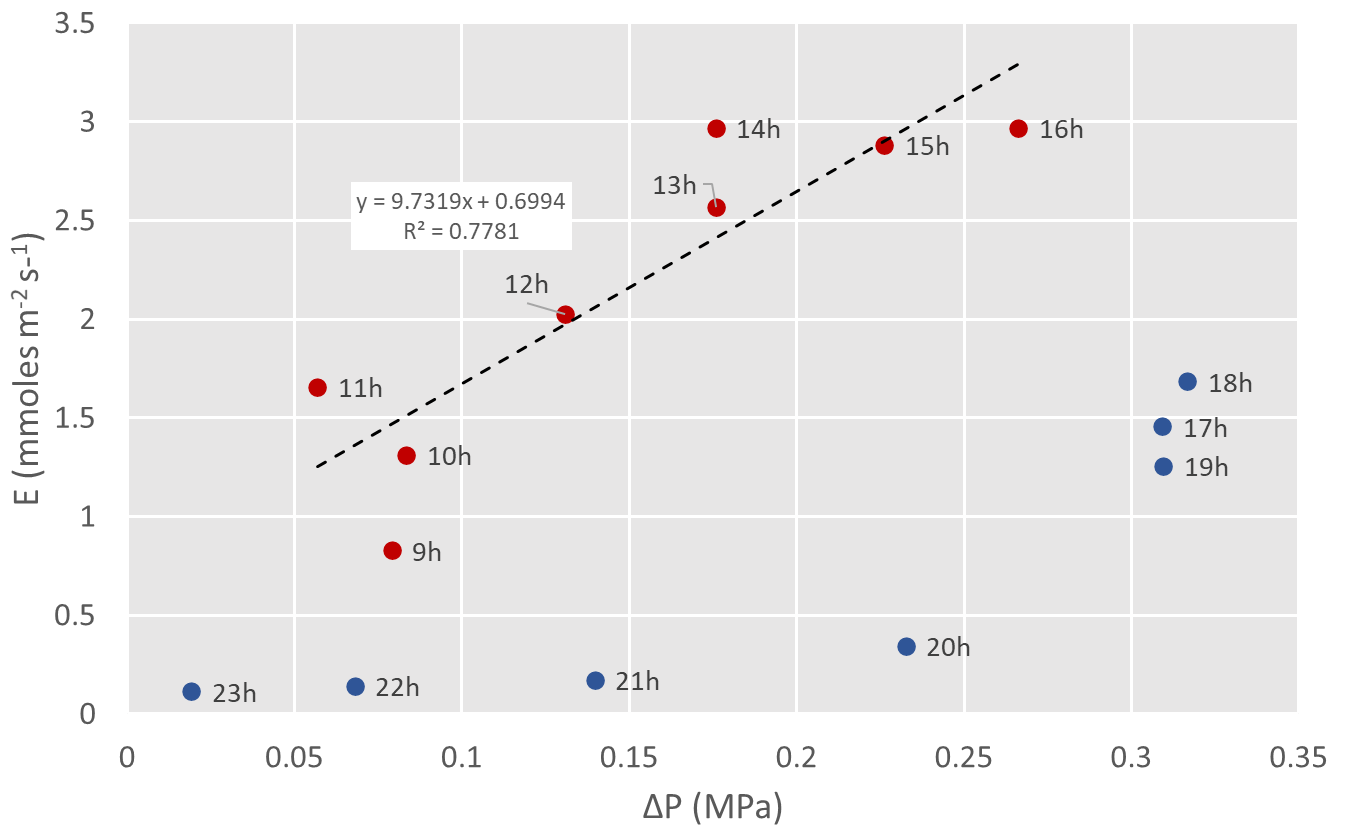
Figure 2: Theoretical example for using diurnal pressure flux relationships to determine plant capacitance.
Conclusion
The longevity of the Scholander pressure chamber is a testament to both its ingenuity, but equally the difficulty of developing tools that move beyond it. So can micro-tensiometers move us beyond the Scholander pressure chamber? I think the answer to this question is clearly no. No single tool is without limitations and the diverse scenarios in which water status must be characterized (different organs, timescales, etc.) means that a variety of tools are needed. Micro-tensiometers have the potential to be a promising addition to the water status toolbox, though their adoption requires reconciling their particular advantages, disadvantages, and lingering questions. One thing appears certain: automated, near real-time Ψ measurements will likely be an critical tool for advancing water relations in the future.
References
- Blanco, V., & Kalcsits, L. (2021). Microtensiometers Accurately Measure Stem Water Potential in Woody Perennials. Plants, 10(12), 2780. https://doi.org/10.3390/plants10122780
- Blanco, V., & Kalcsits, L. (2023). Long-term validation of continuous measurements of trunk water potential and trunk diameter indicate different diurnal patterns for pear under water limitations. Agricultural Water Management, 281, 108257. https://doi.org/10.1016/j.agwat.2023.108257
- Bourbia, I., & Brodribb, T. J. (2023). A new technique for monitoring plant transpiration under field conditions using leaf optical dendrometry. Agricultural and Forest Meteorology, 331, 109328. https://doi.org/10.1016/j.agrformet.2023.109328
- Boyer, J. S. (1967). Leaf Water Potentials Measured with a Pressure Chamber. Plant Physiology, 42(1), 133–137. https://doi.org/10.1104/pp.42.1.133
- Caldeira, C. F., Jeanguenin, L., Chaumont, F., & Tardieu, F. (2014). Circadian rhythms of hydraulic conductance and growth are enhanced by drought and improve plant performance. Nature Communications, 5, 5365. https://doi.org/10.1038/ncomms6365
- Canny, M. (1997). Vessel contents during transpiration—Embolisms and refilling. American Journal of Botany, 84(9), 1223.
- Christenson, C. G., Gohardoust, M. R., Calleja, S., Thorp, K. R., Tuller, M., & Pauli, D. (2024). Monitoring cotton water status with microtensiometers. Irrigation Science, 42(5), 995–1011. https://doi.org/10.1007/s00271-024-00930-w
- Clearwater, M. J., Meinzer, F. C., Andrade, J. L., Goldstein, G., & Holbrook, N. M. (1999). Potential errors in measurement of nonuniform sap flow using heat dissipation probes. Tree Physiology, 19(10), 681–687. https://doi.org/10.1093/treephys/19.10.681
- Cochard, H., Bodet, C., Améglio, T., & Cruiziat, P. (2000). Cryo-scanning electron microscopy observations of vessel content during transpiration in walnut petioles. Facts or artifacts? Plant Physiology, 124(3), 1191–1202. https://doi.org/10.1104/pp.124.3.1191
- Cochard, H., Forestier, S., & Améglio, T. (2001). A new validation of the Scholander pressure chamber technique based on stem diameter variations. Journal of Experimental Botany, 52(359), 1361–1365. https://doi.org/10.1093/jexbot/52.359.1361
- Cohen, Y., Alchanatis, V., Meron, M., Saranga, Y., & Tsipris, J. (2005). Estimation of leaf water potential by thermal imagery and spatial analysis*. Journal of Experimental Botany, 56(417), 1843–1852. https://doi.org/10.1093/jxb/eri174
- Conesa, M. R., Conejero, W., Vera, J., & Ruiz-Sánchez, M. C. (2023). Assessment of trunk microtensiometer as a novel biosensor to continuously monitor plant water status in nectarine trees. Frontiers in Plant Science, 14. https://doi.org/10.3389/fpls.2023.1123045
- Cozzolino, D. (2017). The role of near-infrared sensors to measure water relationships in crops and plants. Applied Spectroscopy Reviews, 52(10), 837–849. https://doi.org/10.1080/05704928.2017.1331446
- De Bei, R., Cozzolino, D., Sullivan, W., Cynkar, W., Fuentes, S., Dambergs, R., Pech, J., & Tyerman, S. (2011). Non-destructive measurement of grapevine water potential using near infrared spectroscopy. Australian Journal of Grape and Wine Research, 17(1), 62–71. https://doi.org/10.1111/j.1755-0238.2010.00117.x
- Dell’acqua, N., Gambetta, G. A., Bartlett, M. K., Burlett, R., Chambard, M., Delzon, S., Ferrer, N., Pinol Daubisse, M., Sinclair, G., & Delmas, C. E. L. (2025). The reduced canopy area in esca-symptomatic grapevine plants leads to lower canopy transpiration and mitigates water stress. Plant Physiology, kiaf361. https://doi.org/10.1093/plphys/kiaf361
- Delzon, S., Sartore, M., Granier, A., & Loustau, D. (2004). Radial profiles of sap flow with increasing tree size in maritime pine. Tree Physiology, 24(11), 1285–1293. https://doi.org/10.1093/treephys/24.11.1285
- Di Biase, R., Calabritto, M., Mininni, A. N., Montanaro, G., & Dichio, B. (2025). Microtensiometer-based trunk water potential as a plant water status indicator in kiwifruit under different soil water availability. Irrigation Science, 43(4), 937–954. https://doi.org/10.1007/s00271-025-01020-1
- Dzikiti, S., Steppe, K., Lemeur, R., & Milford, J. R. (2007). Whole-tree level water balance and its implications on stomatal oscillations in orange trees [Citrus sinensis (L.) Osbeck] under natural climatic conditions. Journal of Experimental Botany, 58(7), 1893–1901. https://doi.org/10.1093/jxb/erm023
- Grimes, D. W., & Yamada, H. (1982). Relation of Cotton Growth and Yield to Minimum Leaf Water Potential. Crop Science, 22(1), cropsci1982.0011183X002200010031x. https://doi.org/10.2135/cropsci1982.0011183X002200010031x
- Haberstroh, S., Scarpa, F., Seeger, S., Christen, A., & Werner, C. (2025). Continuous Stem Water Potential Measurements of a Diffuse-Porous Tree Species Offer New Insights Into Tree Water Relations. Ecohydrology, 18(1), e2761. https://doi.org/10.1002/eco.2761
- Henzler, T., Waterhouse, R., Smyth, A., Carvajal, M., Cooke, D., Schaffner, A., Steudle, E., & Clarkson, D. (1999). Diurnal variations in hydraulic conductivity and root pressure can be correlated with the expression of putative aquaporins in the roots of lotus japonicus. Planta, 210(1), 50–60.
- Lakso, A. N., Santiago, M., & Stroock, A. D. (2022). Monitoring Stem Water Potential with an Embedded Microtensiometer to Inform Irrigation Scheduling in Fruit Crops. Horticulturae, 8(12), 1207. https://doi.org/10.3390/horticulturae8121207
- Meinzer, F. C., James, S. A., Goldstein, G., & Woodruff, D. (2003). Whole-tree water transport scales with sapwood capacitance in tropical forest canopy trees. Plant, Cell & Environment, 26(7), 1147–1155. https://doi.org/10.1046/j.1365-3040.2003.01039.x
- Mirás-Avalos, J. M., & Araujo, E. S. (2021). Optimization of Vineyard Water Management: Challenges, Strategies, and Perspectives. Water, 13(6), Article 6. https://doi.org/10.3390/w13060746
- Moriana, A., Pérez-López, D., Prieto, M. H., Ramírez-Santa-Pau, M., & Pérez-Rodriguez, J. M. (2012). Midday stem water potential as a useful tool for estimating irrigation requirements in olive trees. Agricultural Water Management, 112, 43–54. https://doi.org/10.1016/j.agwat.2012.06.003
- Pagay, V. (2022). Evaluating a novel microtensiometer for continuous trunk water potential measurements in field-grown irrigated grapevines. Irrigation Science, 40(1), 45–54. https://doi.org/10.1007/s00271-021-00758-8
- Pagay, V., Santiago, M., Sessoms, D. A., Huber, E. J., Vincent, O., Pharkya, A., Corso, T. N., Lakso, A. N., & Stroock, A. D. (2014). A microtensiometer capable of measuring water potentials below −10 MPa. Lab on a Chip, 14(15), 2806–2817. https://doi.org/10.1039/C4LC00342J
- Prytz, G., Futsaether, C. M., & Johnsson, A. (2003). Self-sustained oscillations in plant water regulation: Induction of bifurcations and anomalous rhythmicity. New Phytologist, 158(2), 259–267. https://doi.org/10.1046/j.1469-8137.2003.00742.x
- Richards, L. A., & Ogata, G. (1958). Thermocouple for Vapor Pressure Measurement in Biological and Soil Systems at High Humidity. Science, 128(3331), 1089–1090. https://doi.org/10.1126/science.128.3331.1089
- Rodriguez-Dominguez, C. M., Carins-Murphy, M. R., Sebastian-Azcona, J., & Brodribb, T. J. (2025). Stem water potential measurements obtained using standard methodologies diverge under extreme drought in tomato and grapevine. Plant Physiology, 199(4), kiaf591. https://doi.org/10.1093/plphys/kiaf591
- Rodriguez-Dominguez, C. M., Forner, A., Martorell, S., Choat, B., Lopez, R., Peters, J. M. R., Pfautsch, S., Mayr, S., Carins-Murphy, M. R., McAdam, S. A. M., Richardson, F., Diaz-Espejo, A., Hernandez-Santana, V., Menezes-Silva, P. E., Torres-Ruiz, J. M., Batz, T. A., & Sack, L. (2022). Leaf water potential measurements using the pressure chamber: Synthetic testing of assumptions towards best practices for precision and accuracy. Plant, Cell & Environment, 45(7), 2037–2061. https://doi.org/10.1111/pce.14330
- Scholander, P. F., Hammel, H. T., Bradstreet, E. D., & Hemmingsen, E. A. (1965). Sap Pressure in Vascular Plants. Science, 148(3668), 339–346.
- Spanner, D. C. (1951). The Peltier Effect and its Use in the Measurement of Suction Pressure. Journal of Experimental Botany, 2(2), 145–168. https://doi.org/10.1093/jxb/2.2.145
- Torres-Ruiz, J. M., Jansen, S., Choat, B., McElrone, A. J., Cochard, H., Brodribb, T. J., Badel, E., Burlett, R., Bouche, P. S., Brodersen, C. R., Li, S., Morris, H., & Delzon, S. (2015). Direct x-ray microtomography observation confirms the induction of embolism upon xylem cutting under tension. Plant Physiology, 167(1), 40–43. https://doi.org/10.1104/pp.114.249706
- Vandeleur, R. K., Mayo, G., Shelden, M. C., Gilliham, M., Kaiser, B. N., & Tyerman, S. D. (2009). The role of plasma membrane intrinsic protein aquaporins in water transport through roots: Diurnal and drought stress responses reveal different strategies between isohydric and anisohydric cultivars of grapevine. Plant Physiology, 149(1), 445–460. https://doi.org/10.1104/pp.108.128645
- Villalobos, F. J., Testi, L., García-Tejera, O., López-Bernal, Á., Tejado, I., & Vinagre, B. M. (2025). Measuring the Diurnal Variation of Root Conductance in Olive Trees Using Microtensiometers and Sap Flow Sensors. Plant and Soil, 509(1), 999–1012. https://doi.org/10.1007/s11104-024-06873-7
- Wallach, R., Da-Costa, N., Raviv, M., & Moshelion, M. (2010). Development of synchronized, autonomous, and self-regulated oscillations in transpiration rate of a whole tomato plant under water stress. Journal of Experimental Botany, 61(12), 3439–3449. https://doi.org/10.1093/jxb/erq168
- Wei, C., Tyree, M. T., & Steudle, E. (1999). Direct Measurement of Xylem Pressure in Leaves of Intact Maize Plants. A Test of the Cohesion-Tension Theory Taking Hydraulic Architecture into Consideration1. Plant Physiology, 121(4), 1191–1205. https://doi.org/10.1104/pp.121.4.1191
- Ziegler, Y., Grote, R., Alongi, F., Knüver, T., & Ruehr, N. K. (2024). Capturing drought stress signals: The potential of dendrometers for monitoring tree water status. Tree Physiology, 44(12), tpae140. https://doi.org/10.1093/treephys/tpae140
- Zimmermann, D., Reuss, R., Westhoff, M., Geßner, P., Bauer, W., Bamberg, E., Bentrup, F.-W., & Zimmermann, U. (2008). A novel, non-invasive, online-monitoring, versatile and easy plant-based probe for measuring leaf water status. Journal of Experimental Botany, 59(11), 3157–3167. https://doi.org/10.1093/jxb/ern171
- Zimmermann, U., Meinzer, F. C., Benkert, R., Zhu, J. j., Schneider, H., Goldstein, G., Kuchenbrod, E., & Haase, A. (1994). Xylem water transport: Is the available evidence consistent with the cohesion theory? Plant, Cell & Environment, 17(11), 1169–1181. https://doi.org/10.1111/j.1365-3040.1994.tb02015.x
Article statistics
 Views: 493
Views: 493
Downloads
 PDF: 91
PDF: 91
 XML: 23
XML: 23
