Causes of variation in leaf-level drought tolerance within an Amazonian forest
Abstract
Amazonian tree communities have already been seriously impacted by extreme natural droughts, and intense droughts are predicted to increase in frequency. However, our current knowledge of Amazonian tree species’ responses to water stress remains limited, as plant trait databases include few drought tolerance traits, impeding the application and predictive power of models. Here we explored how leaf water potential at turgor loss point (πtlp), a determinant of leaf drought tolerance, varies with species life history, season, tree size and irradiance within a forest in French Guiana. First, we provided a further direct validation of a rapid method of πtlp determination based on osmometer measurements of leaf osmotic potential at full hydration for five Amazonian tree species. Next, we analysed a dataset of 131 πtlp values for a range of species, seasons, size (including saplings), and leaf exposure. We found that early-successional species had less drought-tolerant leaves than late-successional species. Species identity was the major driver of πtlp variation, whereas season, canopy tree size and leaf exposure explained little variation. Shifts in πtlp from saplings to canopy trees varied across species, and sapling leaf drought tolerance was a moderate predictor of canopy tree leaf drought tolerance. Given its low within-species variability, we propose that πtlp is a robust trait, and is useful as one index of species’ drought tolerance. We also suggest that measuring this trait would considerably advance our knowledge on leaf drought tolerance in hyperdiverse communities and would thus likely shed light on the resilience of such vulnerable species-rich ecosystem.
Introduction
Extreme natural droughts have already impacted on Amazonian ecosystem processes and services, through a decrease in tree growth and an increase in tree mortality (Phillips et al. 2009; Lewis et al. 2011; Marengo et al. 2011; Saatchi et al. 2013). In the future, such intense droughts are predicted to increase in frequency over Amazonia (Joetzjer et al. 2013; Boisier et al. 2015). Understanding and predicting the dynamics and responses to drought of Amazonian tree communities thus constitutes a crucial research challenge. We do know that plant water availability is a strong driver of tropical tree species distributions (Engelbrecht et al. 2007; Baltzer et al. 2008; Kursar et al. 2009; Comita & Engelbrecht 2009; Bartlett et al. 2012b; Fauset et al. 2012; Condit et al. 2013; Bartlett et al. 2015). However, our current knowledge of individual species’ responses to water stress remains limited.
Plant functional traits are being increasingly used to explore the processes of plant community assembly (Kraft et al. 2008; Paine et al. 2011; Adler et al. 2013). Traits, such as leaf mass per area, wood density or seed size, are often used as predictors of tree growth trajectories and competitive intensity (Hérault et al. 2011; Lasky et al. 2015 ; but see Paine et al. 2015). Also plant traits play an increasing role in the development of dynamic vegetation models (Scheiter et al. 2013; Fyllas et al. 2014; Sakschewski et al. 2015). Global plant trait databases based on standardized protocols have been instrumental in such approaches (Cornelissen et al. 2003; Chave et al. 2009; Kattge et al. 2011; Pérez-Harguindeguy et al. 2013). However these databases include limited information about the hydraulic functioning of plants, reflecting a historical bias of functional ecology towards the understanding of photosynthesis, wood mechanics, or plant architecture. A major recent effort to compile a stem hydraulic trait database has already highlighted the global vulnerability of forest trees to drought (Choat et al. 2012). Such trait data are essential, as there is growing evidence that often measured plant traits, such as leaf mass per area or wood density, correlate poorly with plant hydraulic responses or drought tolerance (Sack et al. 2003; Sack et al. 2013; Li et al. 2015; Gleason et al. 2015; Maréchaux et al. 2015), though they may be related within given communities, e.g., for drought-sensitive pioneers versus drought-tolerant late-successional species within given forests (Markesteijn et al. 2011a; Markesteijn et al. 2011b). This situation impedes the application and predictive power of trait-based modelling (Wright et al. 2010; Wagner et al. 2014; Paine et al. 2015). Here, we contribute to filling this gap by exploring how the leaf water potential at wilting point, a classical leaf drought tolerance trait, varies across species, seasons, sizes and leaf exposure, in a tropical forest.
The leaf water potential at turgor loss point (πtlp, in MPa) is the negative water potential at which the leaf wilts. It also corresponds to the soil water potential below which the plant cannot take up sufficient water to recover overnight from wilting after a day of transpiration. Leaves with a more negative πtlp maintain critical processes such as leaf hydraulic conductance under drier conditions, contributing to greater plant drought tolerance (Cheung et al. 1975; Abrams et al. 1990; Brodribb & Holbrook 2003; Bartlett et al. 2012b; Guyot et al. 2012). πtlp is a good predictor of species distributions relative to water supply (Lenz et al. 2006; Bartlett et al. 2012b; Bartlett et al. 2015). Recently, we found that tropical forest tree assemblages display substantial species variation in πtlp, with some species being surprisingly drought-tolerant (Nepstad et al. 2007; da Costa et al. 2010; Maréchaux et al. 2015). This study relied on a new protocol for πtlp determination that is an order of magnitude faster than the classic pressure-volume curve method (Bartlett et al. 2012a), based on a well-established correlation of πtlp with the leaf osmotic potential at full hydration, measurable with an osmometer (Sack et al. 2003; Lenz et al. 2006; Blackman et al. 2010; Scoffoni et al. 2011; Bartlett et al. 2012b). Here, we provide the first direct validation of this rapid method of πtlp determination for Amazonian tree species.
The potential associations of the leaf water potential at wilting point with other ecological factors than species have seldom been explored (Choat et al. 2007). Working within a diverse Amazonian forest in French Guiana, we sought to resolve the role of the following ecological factors, which have been hypothesized but not previously tested to drive the variation in leaf drought tolerance, at multiple scales, including across species, within species across individuals, and within individual crowns. Across species, early-successional species have been predicted to show trait values that maximize resource gain at the expense of stress tolerance (Markesteijn et al. 2011a; Markesteijn et al. 2011b; Reich 2014), leading us to expect these species would have less negative πtlp values than late-successional species (hypothesis i). Seasonal variation in water supply is a major driver of intraspecific variation in πtlp for species from diverse ecosystems worldwide, with osmotic adjustment, or the accumulation of cell solutes, acting across time scales from days to seasons to acclimate πtlp to more negative values during water stress (Bartlett et al. 2014). This drought-tolerance mechanism has never been explored for Amazonian tree species, but we expected to see significant seasonal changes in πtlp for our study species (ii). In closed-canopy tropical forests, intraspecific trait variation is often driven by the strong light gradient within the canopy (Niinemets 2010). Emergent trees withstand greater heat and excess light stress and higher evaporative demand leading us to predict that drought tolerance would vary across tree sizes, with larger canopy trees showing more negative πtlp values than smaller canopy trees (iii), and canopy trees showing more negative πtlp values than saplings of the same species (iv). Consistent with hypothesis iii, some studies reported tall trees to be more vulnerable to drought than smaller trees (Van Nieuwstadt & Sheil 2005; Nepstad et al. 2007; da Costa et al. 2010; Phillips et al. 2010; Rowland et al. 2015), although others did not (Enquist & Enquist 2011; Feeley et al. 2011; Fauset et al. 2012). Disentangling the potential structural and physiological causes of these opposed trends has proven difficult (Niinemets 2010). To our knowledge, variation in πtlp has never been compared between mature trees and saplings. Canopy trees often include both shade and sun leaves, the latter being more exposed to heat light and evaporative stress and differing predictably in a number of structural and physiological traits (Sack et al. 2003; Rozendaal et al. 2006; Sack et al. 2006; Markesteijn et al. 2007). We thus predicted sun leaves would show greater drought tolerance, and thus more negative πtlp values than shade leaves within the crown of a single tree (v).
Material and methods
Study sites and sampling strategy
Field measurements were conducted at the Nouragues Ecological Research Station in French Guiana. It is located 120 km south of Cayenne within an undisturbed forest, ca. 50 km from Cacao, the closest village (4°05’ N, 52°40’ W; Bongers et al. 2001). The site receives ca. 3000 mm/yr rainfall, with significant seasonal and inter-annual variation due to the movement of the Inter-Tropical Convergence Zone. A long wet season lasts from December to July, often interrupted by a short dry period in March. The dry season lasts from the end of August to November with 2-3 months with precipitation <100 mm/mo (Bongers et al. 2001). Data were collected in a 12-ha permanent forest plot on clay-sand soil (Petit Plateau). The permanent plot was fully censused in 2012, all trees ≥10 cm diameter at breast height (DBH) were identified and their DBH measured. Within the 12-ha plot, richness is in excess of 600 tree species (C Baraloto, J Engel, P Pétronelli, unpublished results).
Measurements of leaf water potential at turgor loss point (πtlp) were conducted in September 2012 and September 2015 during two dry seasons, and in May 2014 in the middle of the wet season (Appendix S1). The cumulative rainfall 30 days prior the ending date of each sampling session, based on logging half-hourly data at the site (Campbell Scientific SBS500, Shepshed, Leicestershire, UK), was 21 mm, 78 mm and 265 mm for the 2012 dry season, the 2015 dry season, and the 2014 wet season respectively. Ten focal species were selected that were relatively abundant in the study plot to maximize the range in πtlp (Maréchaux et al. 2015). Three to eleven canopy trees were sampled per species in each season (Table 1). Small branches were collected by climbing the trees using French spikes (Fonderies Lacoste, Excideuil, France; de Castilho et al. 2006) or using the single-rope technique (Anderson et al. 2015). For 19 of the 56 canopy trees sampled during the wet season, both sun and shade leaves could be sampled. For five of the ten focal species, saplings (individuals with a height typically less than 1.50m, thus growing in the understory) could be easily identified, and three to seven saplings were sampled per species during the wet season in May 2014 (n=24 saplings total; Table 1).
Estimating πtlp using the osmometer method
We assessed the leaf turgor loss point, πtlp, from a previously established linear relationship with the osmotic potential at full hydration (πo), in turn directly measured with a vapour pressure osmometer (Vapro 5520, Wescor, Logan, UT; Bartlett et al. 2012a). Henceforth, we refer to this technique as the osmometer method.
Branches were collected from the selected trees and placed in opaque bags with wet paper towel, then recut under water at least two nodes distal to the original cut and allowed to rehydrate overnight covered with plastic, and branch ends underwater. Three mature leaves were collected from each rehydrated branch and stored in Whirl-Pak bags (Nasco, Fort Atkinson, WI, USA) with a wet paper towel. One disc was sampled from each leaf centrally between the midrib and margin with an 8-mm-diameter cork borer, excluding first- and second-order veins to avoid too much apoplastic dilution that would lead to less negative osmometer values (Kikuta & Richter 1992). However, for one focal species, Qualea rosea (Vochysiaceae), a dense secondary venation disallowed excluding them. The discs were wrapped in foil and frozen by immersion in liquid nitrogen (LN2) for at least 2 minutes, then immediately punctured 10-15 times with sharp-tipped forceps and sealed in the osmometer chamber, using the standard 10 mL chamber well. The discs were exposed to air for less than 40 seconds for all steps between removing the leaf from the Whirl-Pak bag and sealing the disc in the osmometer. The equilibrium solute concentration value c0 (in mmol kg-1) was recorded from the osmometer when the difference between consecutive 2-minute measurements fell below 5 mmol kg-1. This value was converted to πo values from the osmometer (πosm) using the van’t Hoff equation relating solute concentration to vapour pressure: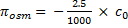 (1)
(1)
where the numerator of the first term represents R´T = 2.5 L MPa mol-1 at 25°C, with R the ideal gas constant and T the temperature in Kelvin degrees.
The value πosm was then converted into πtlp using the previously published regression equation relating πosm to pressure-volume curve turgor loss point values (πtlp-PV; Bartlett et al. 2012a). This regression was developed from woody species representing a wide range of leaf structural features and habitat preferences (n=30 species, R2=86%, p<2.10-12, πosm range=[-3.03, -0.64] MPa and πtlp-PV range=[-3.00, -1.02] MPa) and was: (2)
(2)
This reference regression is based on fundamental equations describing biophysical processes and should therefore hold universally across species. However, we sought to further validate this equation for Amazonian tree species.
Validating the osmometer method for Amazonian tree species
To this end, we calculated the water potential at turgor loss point using the pressure-volume curve method (henceforth denoted the PV curve method; πtlp-PV) and also measured πosm with the osmometer method on the same individuals from six species (n=18 individuals and 6 species). Our validation was conducted in September 2015. Species were excluded if they produce foliar latex or resin, known to create problems with the PV curve method. Branches were allowed to rehydrate overnight as above. Five to six leaves from three individuals per species were dried on a laboratory bench and repeatedly weighed (using a Ohaus Scout Pro Balance SPU 123, Parsippany, NJ, USA) and measured for water potential with a pressure chamber with a digital gauge (PMS Instrument Co., Corvallis, OR, USA). Leaf dry mass was determined after 72 h at 70°C in a drying oven. πtlp-PV was calculated from the relationships between water potential and water content (Koide et al. 2000; Sack et al. 2011). Species-level πtlp-PV was computed as the average of πtlp-PV values of the 5-6 leaves from that species. All πosm and πtlp-PV values are available (Appendix S2).
We compared the root mean squared error (RMSE) from Equation (2) computed for the published calibration dataset (n=30 species; Bartlett et al., 2012a) and for the dataset including the six additional species in this study (n=6 and n=36 species). RMSE was computed as follows: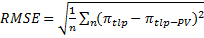 (3)
(3)
where πtlp values were turgor loss point estimates with Equation (2). We identified potential outliers in the merged dataset (n=36 species) using jackknife resampling method with RMSE as statistics (R package bootstrap; Tibshirani & Leisch 2015). We also fitted the 6-species and the merged dataset and compared the 95% confidence intervals of slope and intercept of the 6- and 36-species datasets with the 30-species dataset based on 1000 bootstraps (R package boot; Canty & Ripley 2013).
Data analyses
All analyses were conducted for the wet-season πtlp values (May 2014), except for the comparison between seasons. We considered the mean of the sun and shade leaves πtlp values for individuals for which both were collected, excepted when comparing leaves sampled at different light exposures. Also, for reasons explained below, individuals from one species, Qualea rosea, were excluded from all subsequent tests.
To test for species differences in πtlp values, we used linear models (ANOVA), with species as a fixed effect, and Tukey HSD tests for pairwise species comparisons. The relative contribution of within- and across-species differences to πtlp variability was quantified by partitioning variance (Sokal & Rohlf 1987). We also tested for differences in πtlp between species grouped into early-successional, mid-successional and late-successional species according to field observations (Table 1), using an ANOVA, with successional status as a fixed effect, and Tukey HSD for post hoc pairwise comparisons.
We compared wet-season canopy tree values with dry-season values with the same osmometer method (Maréchaux et al. 2015) for six species with at least three individuals sampled during each season. We tested for a seasonal effect using a two-way ANOVA with season and species as fixed effects and using t-tests for each species.
Using DBH as a proxy for tree size, we investigated the relationships between canopy tree DBH and πtlp, and repeated this analysis accounting for the species effect (linear regressions between DBH and residuals of the ANOVA on πtlp with species as a fixed effect) to exclude variability caused by species differences in stature. We also tested differences in πtlp for four species for which we sampled both canopy trees and saplings, using a two-way ANOVA (type II) with size (categorical: sapling/canopy) and species as fixed effects.
We tested differences in πtlp between leaves collected in full sunlight at top canopy height (henceforth denoted sun leaves) and leaves collected in shaded conditions lower in the canopy (shade leaves) within individuals using a paired t-test (n=18 individuals). We validated our classification of sun and shade leaves by testing for differences in additional traits known to vary with light environments (Niinemets et al. 2015), i.e., leaf mass per area (P<10-4, mean of the differences=15.3 g.m-2), and carbon isotope ratio (P<10-3, mean of differences=1.23 ‰; data not shown).
All statistical analyses were conducted using the R software (R Core Team 2013) and preceded by normality and homoscedasticity tests when required, using Shapiro-Wilk test, Bartlett test or Levene test (car package; Fox & Weisberg 2011).
Results
Model validation with Amazonian tree species
The species Qualea rosea, with its dense secondary vein network was clearly identified as an outlier: for this species, the PV-curve value of πtlp value was much lower than that obtained with the osmometer method (πtlp- πtlp-PV=0.58 MPa). We decided to remove this outlier species from all subsequent analyses. The RMSEs of the 5- and 35-species datasets were both 0.18 MPa, equal to that of the previously constructed 30-species model (Bartlett et al., 2012a). 95% confidence intervals of the slope and the intercept of both of the 5- and 35-species regressions included the parameter estimates for the previous 30-species calibration. The new correlation between πosm and πtlp-PV, established based on the 35 species was: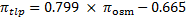 (4)
(4)
(P<10-14, R2=0.85, RMSE=0.18 MPa, Fig. 1). We conclude that this relationship is applicable to Neotropical trees, as assumed previously (Maréchaux et al. 2015), provided the leaf secondary vein network is not too dense.

Figure 1: Validation of the osmometer method for Amazonian tree species. Linear regressions between species leaf water potentials at turgor loss point measured with the PV curve method (πtlp-PV, in MPa) and species osmotic potentials at full hydration measured with an osmometer (πosm, in MPa), for the (Bartlett et al. 2012a) 30 species means (solid line) only and including the five species in this study (excluding Qualea rosea, 35 species, dashed line). The regressions were not significantly different.
Cross-species variation in πtlp
The πtlp of canopy trees varied across species (Table 1, Fig. 2; ANOVA, P<1.10-15), with 87% of the variance in πtlp associated with species differences (n=9 species and 50 individuals). Early-successional (n=2 species and 11 individuals) exhibited significantly less negative πtlp values than mid-successional species (n=2 species and 10 individuals). In turn, mid-successional species exhibited less negative πtlp values than late-successional species (n=5 species and 29 individuals; ANOVA P<1.10-7, Tukey HSD tests: all P<0.05; Fig. 3).
Table 1: Mean and standard error of πtlp values in wet and dry seasons and for saplings and canopy trees.
| WET SEASON | DRY SEASON | |||||||
| Canopy trees | Saplings | Canopy trees | ||||||
| Species | Type | n | πtlp | n | πtlp | n | πtlp | |
| Protium gallicum | LS | 6 | -2.52 ± 0.05 | 3 | -2.29 ± 0.20 | 4 | 2.50 ± 0.10 | |
| Voucapoua americana | LS | 6 | -2.17 ± 0.06c | 6 | -2.15 ± 0.08 | |||
| Goupia glabra | MS | 4 | -2.09 ± 0.03c | 3 | -2.02 ± 0.05 | |||
| Licania alba | LS | 7 | -2.03 ± 0.05c | 6 | -1.83 ± 0.04 | 11 | -2.01 ± 0.04 | |
| Cyrillopsis paraensis | LS | 5 | -2.00 ± 0.03cf | |||||
| Eschweilera coriacea | LS | 5 | -1.76 ± 0.08bef | 11 | -1.76 ± 0.05 | |||
| Pourouma sp. | ES | 3 | -1.54 ± 0.10abd | 3 | -1.76 ± 0.10 | |||
| Dicorynia guianensis | MS | 6 | -1.52 ± 0.04abd | 6 | -1.41 ± 0.03 | 10 | -1.66 ± 0.08 | |
| Cecropia obtusa | ES | 8 | -1.43 ± 0.04a | |||||
ES: early-successional; MS: mid-successional; LS: late-successional.
Same lower case letters indicate a non-significant difference between species values for canopy trees (Tukey test: P>0.05).
Seasonal and size-related variation in πtlp within species
We found no difference between dry- and wet-season πtlp for any of the tested tree species (n=6 species; two-way ANOVA; t-tests: all p-values > 0.3, Table 1, Fig. 2). This was still the case when excluding data collected during the September 2015 sampling session which was less dry than the particularly stressed period of September 2012.
The πtlp was not statistically related to DBH among canopy trees (linear regression, P=0.44, n=50 individuals). Further, DBH did not explain residual πtlp variance among canopy trees after accounting for the species effect (linear regression between DBH and the residuals of ANOVA on πtlp with species as a fixed effect, P=0.43). This result also held when excluding individuals growing in large open gaps (n=7 species and 37 individuals, P=0.48), for which a variation in size may not result in a variation in abiotic stresses due to changes of vapour pressure deficit, temperature or light intensities.
Species, plant stature (canopy vs sapling) and the interaction of these two effects had a significant effect on πtlp (two-way ANOVA, n=4 species, all P≤0.05, range of average πtlp differences between stature within species: [-0.23, -0.11] MPa). For all species, canopy trees displayed a more negative πtlp than saplings, except the early-successional Pourouma sp. which showed the opposite pattern (Table 1, Fig. 2).

Figure 2: Leaf water potential at wilting point (πtlp, in MPa) across species in our dataset, with mean ± standard error for species wet-season canopy tree (black), dry-season canopy tree (dark grey) and wet-season sapling (light grey) values. * indicates significant differences between saplings and canopy trees during the wet season within species (t.test : P<0.05).

Figure 3: Differences in leaf water potential at wilting point (πtlp, in MPa) across successional stages, for early- , mid- and late-successional canopy trees (n=11, 10 and 29 individuals respectively). Means ± standard errors are reported. Different lower case letters indicate a significant difference between stages (Tukey test: P<0.05).
Intra-crown variation in πtlp
Differences between sun and shade leaves within individuals were marginally significant, remaining lower than the model’s RMSE (paired t-test, P=0.05, mean of the differences=-0.09 MPa, 95% confident interval of the difference= [-0.18 ; -0.00] MPa; n=18 individuals; Fig. 4).
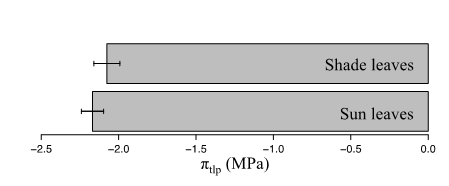
Figure 4: Comparison between sun and shade leaves leaf water potential at wilting point (πtlp, in MPa) for 18 tropical canopy trees, with mean ± standard error.
Discussion
Validation of the osmometer method for Amazonian tree species
We applied a rapid method of leaf turgor loss point determination based on osmometer measurements of leaf osmotic potential at full hydration (πosm). The correlation between πosm and πtlp was initially established for 15 species sampled in gardens adjacent to the campus of University of California, Los Angeles, from vegetation zones ranging from chaparral to tropical wet forest, and a further 15 species growing in natural conditions in a Chinese tropical rainforest (Bartlett et al. 2012a). These two subgroups did not differ statistically in the relationship between measurement methods, consistent with the expectation that because the relationship is biophysically based, it can be applied across vegetation zones (Bartlett et al. 2012a). Here, we included five more species from canopy tropical trees, and confirmed the strong consistency of the correlation, lending further credence to the proposal that it can be applied universally across angiosperm species. Model error remained consistently low after adding the new species. Indeed, the new correlation was not significantly different from the previously established one. We thus conclude that the original model, or its slight improvement reported in Equation (4), can be reliably used to infer leaf-level potential at wilting point across a wide range of angiosperm species.
The rapid method offers invaluable opportunities to better inform and quantify the diversity of leaf drought tolerance in such vulnerable species-rich plant communities. The πtlp has been used as a proxy for general plant drought tolerance, because in several species sets it was notably correlated with specialization of species between wet and dry forests and biomes (Bartlett et al. 2012b) and the water potential at stomatal closure (Brodribb et al. 2003; Brodribb & Holbrook 2003; Blackman et al. 2010), even though more research is required to further elucidate the mechanistic relationships among traits which could potentially arise due to mechanistic linkage and/or to simultaneous co-selection during evolution, e.g., for performance under a given moisture availability regime. Besides, in particular for tropical tree species, the measurement of xylem resistance to cavitation is challenging because of the presence of long vessels (Martin-StPaul et al. 2014; Torres-Ruiz et al. 2014 ; Delzon 2015), and few measurements of the xylem pressure at which 50% loss of conductivity occurs (P50) are available, although these trait was correlated with πtlp in several data sets (Choat et al. 2007; Blackman et al. 2010; Fu et al. 2012). Nine published P50 values for tree genera present at our site were reported in the global database of Choat et al. (2012), including six genera collected at the Parque Nacional San Lorenzo, Panama and three genera at La Selva Research Station, Costa Rica. The range of P50 values ([-2.70; -1.00] MPa) was comparable to the range of πtlp values found at our site (this study: [-2.52; -1.43] MPa and Maréchaux et al. 2015: [-3.15; -1.41] MPa). Since πtlp is currently the only easily measurable drought tolerance trait for tropical woody angiosperms, it is important to further explore the relationship between P50 and πtlp for these species.
One species, Qualea rosea, departed from the model’s predictions. This species displayed a dense network of leaf secondary veins, and the osmometer method led to an over-estimated (less-negative) turgor loss point value. This is consistent with previous studies finding that including secondary veins in a sample leads to a greater apoplastic dilution and thus less negative osmometer measurements (Kikuta & Richter 1992). Species with very dense secondary vein networks should be carefully excluded when using this method. In the French Guiana flora, the number of species with dense secondary venation is limited: only about 2.5% of the tree genera, and <2% of tree species contain species with a dense secondary venation (J Engel, personal communication).
Variation in πtlp strongly determined by species identity and life history
Species identity was the major determinant of leaf drought tolerance, and inter-specific variation in πtlp was larger than intra-specific or intra-individual variation. In contrast, season, canopy tree size or leaf light exposure did not explain much of this variability.
Species identity explained 87% of the variance of πtlp in our dataset (n=9 species, 50 individuals), a larger proportion than previously found (40 %, n=13 species, 82 individuals; Maréchaux et al., 2015). This result is in agreement with previous studies in other ecosystem types (Lenz et al., 2006; Bartlett et al., 2015). Overestimation of interspecific variation in plant traits may result from standardized protocols minimizing intraspecific variation (Cornelissen et al. 2003; Violle et al. 2012). However, this alone could not explain our pattern since our leaf sampling was not standardized with respect to light exposure or canopy tree size and was designed to maximise the entire range of the selected species. We acknowledge that we sampled species at a single site which may induce a lower intra-specific variability than if we had sampled across many sites (Hulshof & Swenson 2010; Albert et al. 2010; Siefert et al. 2015). However in a previous study, variation as assessed at two sites did not result in a greater intraspecific variation in πtlp (Maréchaux et al. 2015).
As hypothesized from a whole-plant “fast-slow” economics spectrum (Reich 2014) and reported coordination of species’ drought and shade tolerance (Markesteijn et al. 2011a; Markesteijn et al. 2011b), early-successional species tended to exhibit less negative πtlp values than later-stage species. This is in agreement with light-demanding species showing higher maximum stomatal conductance and assimilation and transpiration rates than shade-tolerant species under well-watered conditions, but a stronger sensitivity of these variables to soil drought, probably due to a more sensitive stomatal control on average in early-successional species (Huc et al. 1994; Bonal et al. 2000).
Absence of seasonal osmotic adjustment
We found no seasonal adjustment in πtlp for the five species with both dry and wet seasons values in our dataset. This result reinforces the view that some tree species in French Guiana are far more drought-tolerant than previously assumed for moist tropical forests, perhaps due to adaptation to past and recurring drought events in the region (Maréchaux et al. 2015). This finding runs contrary to what was expected from a global meta-analysis that reported consistent and prevalent seasonal plasticity in πtlp across 246 species from eight biomes worldwide (Bartlett et al. 2014). However, that meta-analysis also concluded that there is a wide variability in plasticity across species. The few existing studies that reported tropical moist species πtlp values across seasons provided mixed evidence for osmotic adjustment in tropical tree leaves (Wright et al. 1992; Cao 2000; Brodribb et al. 2003).
One reason explaining the absence of osmotic adjustment in our dataset could be that dry seasons rarely result in a strong tree physiological stress in French Guiana. Values of predawn and midday leaf water potentials measured during an average dry season (Stahl et al. 2011; Stahl et al. 2013) usually were less negative than πtlp values reported at the same site (Maréchaux et al. 2015): at the end of the dry season in November 2007, the range of predawn leaf water potentials were [-1.30;-0.15] MPa, 75% of which were less negative than -0.45 MPa (n=64 individuals), while the range for midday leaf water potentials were [-2.95;-0.60] MPa, 75% of which were less negative than -1.90 MPa (n=49 individuals; C Stahl personal communication). Thus, we do not exclude the possibility of osmotic adjustment under harsher droughts.
πtlp does not depend on canopy tree size, but differs for saplings
In agreement with our prediction, we found that canopy tree size did not have a significant effect on πtlp, whether through a species effect or within species. This finding is interesting in light of studies that have suggested a higher vulnerability to drought of large trees compared with smaller trees (Van Nieuwstadt & Sheil 2005; Nepstad et al. 2007; da Costa et al. 2010; Phillips et al. 2010; Bennett et al. 2015), although this pattern was not observed in other studies (Enquist & Enquist 2011; Feeley et al. 2011; Fauset et al. 2012). Emergent trees are indeed exposed to greater atmospheric evaporative demand and hydraulic constraints. However, these large trees can also store larger amounts of water and thus withstand prolonged periods of stomatal closure (Goldstein et al. 1998; Meinzer et al. 2008), compete strongly for soil resources with their large root systems and access the water table deeper down (Nepstad et al. 1994; Dawson 1996; Ivanov et al. 2012; but see Meinzer et al. 1999; Markewitz et al. 2010; Stahl, Herault, et al. 2013), and accumulate more non-structural carbohydrates that may help maintain high osmotically active compound concentrations and thus attenuate the effect of drought (Sala & Hoch 2009; Woodruff & Meinzer 2011; Sala et al. 2012; O’Brien et al. 2014 ; but see Rowland et al. 2015).
Our results indicate that tall tropical trees do not have a higher leaf-level drought tolerance relative to smaller trees. This in turn suggests that large trees are not more vulnerable to drought than smaller trees. However we found evidence for a weak shift in leaf drought tolerance between saplings and canopy trees. Notably, early- and late-successional species showed opposed trends in their variation in πtlp between saplings and trees, although not all were significant in our dataset of limited sample size. In the late-successional species, leaves of saplings were less drought-tolerant than those of canopy trees. In contrast, for species growing in gaps, leaves were more drought-tolerant at the sapling stage. Such contrasting shifts from saplings to canopy trees across species suggest that these species operate under differential ecophysiological constraints. Differential ability to cope with drought across plant sizes has been reported in relation to changes in environmental constraints (Donovan & Ehleringer 1991; Donovan & Ehleringer 1992; Cavender-Bares & Bazzaz 2000; Niinemets 2010). Soil water availability also differs between gaps and the understory: gaps are more controlled by the evaporation of soil surface while closed-canopy forest soil water is depleted by dense root water extraction below the surface (Becker et al. 1988; Marthews et al. 2008). Saplings of early-successional species, growing in gaps, with typically more superficial roots than taller individuals despite similar high-light exposure, may thus be strongly stressed even during short dry spells in the wet season (Engelbrecht et al. 2006). On the contrary late-successional saplings, growing in closed understory, may be more strongly impacted by herbivory and thus invest more in leaf defense against herbivores than in drought tolerance (Coley & Barone 1996). It would be useful to increase sample size to further analyse these trends, as the contribution of shifts in πtlp to defining the ecological niche of plant species seems to be a promising research avenue.
A weak variation in πtlp across leaf exposure within tree canopy
Sun leaves showed structural and physiological acclimation to high radiation loads and high vapour pressure deficit, with typically higher mass per area and nutrient concentration per area than shade leaves, as expected from previous studies (Sack et al. 2003; Rozendaal et al. 2006; Sack et al. 2006; Markesteijn et al. 2007; Hulshof & Swenson 2010; Weerasinghe et al. 2014). However, we found no substantial differences in absolute πtlp values between sun and shade leaves just as reported previously for temperate woody species (Sack et al. 2003). A strong acclimation in traits related to maximum flux of water and carbon through the leaf, such as leaf hydraulic conductance, vein density, and photosynthetic gas exchange may occur alongside little acclimation in traits associated with leaf drought tolerance (Sack et al. 2003; Sack & Holbrook 2006).
On measuring the community-wide leaf water potential at turgor loss point
The rapid technique of πtlp determination validated here for Amazonian tree species opens new possibilities for studying drought tolerance within and across species. This gives access to a key drought tolerance trait as part of the plant ecologist’s toolkit of routinely measured functional traits (Cornelissen et al. 2003). We propose that it should now be possible to survey the πtlp in hyperdiverse communities and thus shed light on both the resilience of this ecosystem and on how the Amazonian flora has been assembled.
We found that species identity was the main driver of πtlp values with early-successional species being less drought-tolerant than later-successional species, and that season, canopy tree size and leaf exposure contributed relatively negligible variation. Yet, we explored only some of the factors that might affect inter-individual variability, species plasticity, and species differences. For instance we did not study the effect of topography (Silva et al. 2013; Liu et al. 2014) or species maximum height (Poorter et al. 2005). Intra-specific variability, whatever the drivers, may have direct consequences on sampling design and effort for community-wide measurements. However, our result confirms that πtlp could be used as a suitable species-level trait of tree drought tolerance, thus considerably simplifying sampling and determination of drought tolerance in species-rich communities (Baraloto et al. 2010).
We also provided evidence of an ontogenetic shift in πtlp that is variable across species. Particular care should thus be taken when scaling to canopy trees leaf-level drought tolerance measurements based only on saplings (Engelbrecht et al. 2007; Kursar et al. 2009). More research is needed to elucidate how such a variation across ontogeny and across species may impact species performance and vital rates across the life cycle (Adler et al. 2014) and under multiple stresses (Niinemets 2010).
Acknowledgments: we thank V Alt and S Counil for their help in tree climbing and R Gaspard and A Granchamp for their help in the field. This work has benefitted from ‘Investissement d’Avenir’ grants managed by the French Agence Nationale de la Recherche (CEBA, ref. ANR-10-LABX-25-01 and TULIP, ref. ANR-10-LABX-0041; ANAEE-France: ANR-11-INBS-0001) and from additional funds (ANR BRIDGE project; CNRS).
References
- Abrams M, Kubiske M, Steiner K. 1990. Drought adaptations and responses in 5 genotypes of Fraxinus pennsylvanica Marsh.: photosynthesis, water relations and leaf morphology. Tree Physiology 6: 305–315. doi:10.1093/treephys/6.3.305
- Adler PB, Fajardo A, Kleinhesselink AR, Kraft NJB. 2013. Trait-based tests of coexistence mechanisms. Ecology Letters 16: 1294–1306. doi:10.1111/ele.12157
- Adler PB, Salguero-Gómez R, Compagnoni A, Hsu JS, Ray-Mukherjee J, Mbeau-Ache C, Franco M. 2014. Functional traits explain variation in plant life history strategies. Proceedings of the National Academy of Sciences 111: 740–745. doi:10.1073/pnas.1315179111
- Albert CH, Thuiller W, Yoccoz NG, Douzet R, Aubert S, Lavorel S. 2010. A multi-trait approach reveals the structure and the relative importance of intra- vs. interspecific variability in plant traits. Functional Ecology 24: 1192–1201. doi:10.1111/j.1365-2435.2010.01727.x
- Anderson DL, Koomjian W, French B, Altenhoff SR, Luce J. 2015. Review of rope-based access methods for the forest canopy: safe and unsafe practices in published information sources and a summary of current methods. Methods in Ecology and Evolution 6: 865–872. doi:10.1111/2041-210X.12393
- Baltzer JL, Davies SJ, Bunyavejchewin S, Noor NSM. 2008. The role of desiccation tolerance in determining tree species distributions along the Malay–Thai Peninsula. Functional Ecology 22: 221–231. doi:10.1111/j.1365-2435.2007.01374.x
- Baraloto C, Paine CET, Patiño S, Bonal D, Hérault B, Chave J. 2010. Functional trait variation and sampling strategies in species-rich plant communities. Functional Ecology 24: 208–216. doi:10.1111/j.1365-2435.2009.01600.x
- Bartlett MK, Scoffoni C, Ardy R, Zhang Y, Sun S, Cao K, Sack L. 2012a. Rapid determination of comparative drought tolerance traits: using an osmometer to predict turgor loss point. Methods in Ecology and Evolution 3: 880–888. doi:10.1111/j.2041-210X.2012.00230.x
- Bartlett MK, Scoffoni C, Sack L. 2012b. The determinants of leaf turgor loss point and prediction of drought tolerance of species and biomes: a global meta-analysis. Ecology Letters 15: 393–405. doi:10.1111/j.1461-0248.2012.01751.x
- Bartlett MK, Zhang Y, Kreidler N, Sun S, Ardy R, Cao K, Sack L. 2014. Global analysis of plasticity in turgor loss point, a key drought tolerance trait. Ecology Letters 17: 1580–1590. doi:10.1111/ele.12374
- Bartlett MK, Zhang Y, Yang J, Kreidler N, Sun S, Lin L, Hu Y-H, Cao K-F, Sack L. 2015. Drought tolerance as a driver of tropical forest assembly: resolving spatial signatures for multiple processes. Ecology doi:10.1890/15-0468.1
- Becker P, Rabenold PE, Idol JR, Smith AP. 1988. Water potential gradients for gaps and slopes in a Panamanian tropical moist forest’s dry season. Journal of Tropical Ecology 4: 173–184. doi:10.1017/S0266467400002674
- Bennett AC, McDowell NG, Allen CD, Anderson-Teixeira KJ. 2015. Larger trees suffer most during drought in forests worldwide. Nature Plants 1: 15139. doi:10.1038/nplants.2015.139
- Blackman CJ, Brodribb TJ, Jordan GJ. 2010. Leaf hydraulic vulnerability is related to conduit dimensions and drought resistance across a diverse range of woody angiosperms. New Phytologist 188: 1113–1123. doi:10.1111/j.1469-8137.2010.03439.x
- Boisier JP, Ciais P, Ducharne A, Guimberteau M. 2015. Projected strengthening of Amazonian dry season by constrained climate model simulations. Nature Climate Change 5: 656–660. doi:10.1038/nclimate2658
- Bonal D, Barigah TS, Granier A, Guehl JM. 2000. Late-stage canopy tree species with extremely low δ13C and high stomatal sensitivity to seasonal soil drought in the tropical rainforest of French Guiana. Plant Cell and Environment 23: 445–459. doi:10.1046/j.1365-3040.2000.00556.x
- Brodribb TJ, Holbrook NM. 2003. Stomatal closure during leaf dehydration, correlation with other leaf physiological traits. Plant Physiology 132: 2166–2173. doi:10.1104/pp.103.023879
- Brodribb TJ, Holbrook NM, Edwards EJ, Gutiérrez MV. 2003. Relations between stomatal closure, leaf turgor and xylem vulnerability in eight tropical dry forest trees. Plant Cell and Environment 26: 443–450. doi:10.1046/j.1365-3040.2003.00975.x
- Canty A, Ripley B. 2013. boot: Bootstrap R (S-Plus) Functions. R package version 1.3-9.
- Cao K. 2000. Water relations and gas exchange of tropical saplings during a prolonged drought in a Bornean heath forest, with reference to root architecture. Journal of Tropical Ecology 16: 101–116. doi:10.1017/S0266467400001292
- de Castilho CV, Magnusson WE, Oliveira de Araújo RN, Da Costa Pereira E, De Souza SS. 2006. The use of French spikes to collect botanical vouchers in permanent plots: evaluation of potential impacts. Biotropica 38:555–557. doi:10.1111/j.1744-7429.2006.00172.x
- Cavender-Bares J, Bazzaz FA. 2000. Changes in drought response strategies with ontogeny in Quercus rubra: implications for scaling from seedlings to mature trees. Oecologia 124: 8–18. doi:10.1007/PL00008865
- Chave J, Coomes D, Jansen S, Lewis SL, Swenson NG, Zanne AE. 2009. Towards a worldwide wood economics spectrum. Ecology Letters 12: 351–366. doi:10.1111/j.1461-0248.2009.01285.x
- Cheung YNS, Tyree MT, Dainty J. 1975. Water relations parameters on single leaves obtained in a pressure bomb and some ecological interpretations. Canadian Journal of Botany 53:1342–1346. doi:10.1139/b75-162
- Choat B, Jansen S, Brodribb TJ, Cochard H, Delzon S, Bhaskar R, Bucci SJ, Feild TS, Gleason SM, Hacke UG, et al. 2012. Global convergence in the vulnerability of forests to drought. Nature 491: 752–755. doi:10.1038/nature11688
- Choat B, Sack L, Holbrook NM. 2007. Diversity of hydraulic traits in nine Cordia species growing in tropical forests with contrasting precipitation. New Phytologist 175:686–698. doi:10.1111/j.1469-8137.2007.02137.x
- Coley PD, Barone JA. 1996. Herbivory and Plant Defenses in Tropical Forests. Annual Review of Ecology and Systematics 27: 305–335. doi:10.1146/annurev.ecolsys.27.1.305
- Comita LS, Engelbrecht BMJ. 2009. Seasonal and spatial variation in water availability drive habitat associations in a tropical forest. Ecology 90: 2755–2765. doi:10.1890/08-1482.1
- Condit R, Engelbrecht BMJ, Pino D, Pérez R, Turner BL. 2013. Species distributions in response to individual soil nutrients and seasonal drought across a community of tropical trees. Proceedings of the National Academy of Sciences 110: 5064–5068. doi:10.1073/pnas.1218042110
- Cornelissen JHC, Lavorel S, Garnier E, Díaz S, Buchmann N, Gurvich DE, Reich PB, Steege H ter, Morgan HD, Heijden MGA van der, et al. 2003. A handbook of protocols for standardised and easy measurement of plant functional traits worldwide. Australian Journal of Botany 51: 335–380. doi:10.1071/BT02124
- da Costa ACL, Galbraith D, Almeida S, Portela BTT, da Costa M, de Athaydes Silva Junior J, Braga AP, de Gonçalves PHL, de Oliveira AA, Fisher R, et al. 2010. Effect of 7 yr of experimental drought on vegetation dynamics and biomass storage of an eastern Amazonian rainforest. New Phytologist 187: 579–591. doi:10.1111/j.1469-8137.2010.03309.x
- Dawson TE. 1996. Determining water use by trees and forests from isotopic, energy balance and transpiration analyses: the roles of tree size and hydraulic lift. Tree Physiology 16: 263–272. doi:10.1093/treephys/16.1-2.263
- Delzon S. 2015. New insight into leaf drought tolerance. Functional Ecology 29: 1247–1249. doi:10.1111/1365-2435.12500
- Donovan LA, Ehleringer JR. 1991. Ecophysiological differences among juvenile and reproductive plants of several woody species. Oecologia 86: 594–597. doi:10.1007/BF00318327
- Donovan LA, Ehleringer JR. 1992. Contrasting Water-Use Patterns Among Size and Life-History Classes of a Semi-Arid Shrub. Functional Ecology 6: 482–488. doi:10.2307/2389287
- Engelbrecht BMJ, Comita LS, Condit R, Kursar TA, Tyree MT, Turner BL, Hubbell SP. 2007. Drought sensitivity shapes species distribution patterns in tropical forests. Nature 447: 80–82. doi:10.1038/nature05747
- Engelbrecht BMJ, Dalling JW, Pearson TRH, Wolf RL, Galvez DA, Koehler T, Tyree MT, Kursar TA. 2006. Short dry spells in the wet season increase mortality of tropical pioneer seedlings. Oecologia 148: 258–269. doi:10.1007/s00442-006-0368-5
- Enquist BJ, Enquist CA. 2011. Long-term change within a Neotropical forest: assessing differential functional and floristic responses to disturbance and drought. Global Change Biology 17: 1408–1424. doi:10.1111/j.1365-2486.2010.02326.x
- Fauset S, Baker TR, Lewis SL, Feldpausch TR, Affum-Baffoe K, Foli EG, Hamer KC, Swaine MD. 2012. Drought-induced shifts in the floristic and functional composition of tropical forests in Ghana. Ecology Letters 15: 1120–1129. doi:10.1111/j.1461-0248.2012.01834.x
- Feeley KJ, Davies SJ, Perez R, Hubbell SP, Foster RB. 2011. Directional changes in the species composition of a tropical forest. Ecology 92: 871–882. doi:10.1890/10-0724.1
- Fox J, Weisberg S. 2011. An {R} Companion to Applied Regression. Thousand Oaks, CA, USA: Sage Publications. Available from: http://socserv.socsci.mcmaster.ca/jfox/Books/Companion
- Fu P-L, Jiang Y-J, Wang A-Y, Brodribb TJ, Zhang J-L, Zhu S-D, Cao K-F. 2012. Stem hydraulic traits and leaf water-stress tolerance are co-ordinated with the leaf phenology of angiosperm trees in an Asian tropical dry karst forest. Annals of Botany 110: 189–199. doi:10.1093/aob/mcs092
- Fyllas NM, Gloor E, Mercado LM, Sitch S, Quesada CA, Domingues TF, Galbraith DR, Torre-Lezama A, Vilanova E, Ramírez-Angulo H, et al. 2014. Analysing Amazonian forest productivity using a new individual and trait-based model (TFS v.1). Geoscientific Model Development 7: 1251–1269. doi:10.5194/gmd-7-1251-2014
- Gleason SM, Westoby M, Jansen S, Choat B, Hacke UG, Pratt RB, Bhaskar R, Brodribb TJ, Bucci SJ, Cao K-F, et al. 2016. Weak tradeoff between xylem safety and xylem-specific hydraulic efficiency across the world’s woody plant species. New Phytologist 209: 123–136. doi:10.1111/nph.13646
- Goldstein G, Andrade JL, Meinzer FC, Holbrook NM, Cavelier J, Jackson P, Celis A. 1998. Stem water storage and diurnal patterns of water use in tropical forest canopy trees. Plant Cell and Environment 21: 397–406. doi:10.1046/j.1365-3040.1998.00273.x
- Guyot G, Scoffoni C, Sack L. 2012. Combined impacts of irradiance and dehydration on leaf hydraulic conductance: insights into vulnerability and stomatal control. Plant Cell and Environment 35: 857–871. doi:10.1111/j.1365-3040.2011.02458.x
- Hérault B, Bachelot B, Poorter L, Rossi V, Bongers F, Chave J, Paine CET, Wagner F, Baraloto C. 2011. Functional traits shape ontogenetic growth trajectories of rain forest tree species. Journal of Ecology 99: 1431–1440. doi:10.1111/j.1365-2745.2011.01883.x
- Huc R, Ferhi A, Guehl JM. 1994. Pioneer and late stage tropical rainforest tree species (French Guiana) growing under common conditions differ in leaf gas exchange regulation, carbon isotope discrimination and leaf water potential. Oecologia 99: 297–305. doi:10.1007/BF00627742
- Hulshof CM, Swenson NG. 2010. Variation in leaf functional trait values within and across individuals and species: an example from a Costa Rican dry forest. Functional Ecology 24: 217–223. doi:10.1111/j.1365-2435.2009.01614.x
- Ivanov VY, Hutyra LR, Wofsy SC, Munger JW, Saleska SR, de Oliveira RC, de Camargo PB. 2012. Root niche separation can explain avoidance of seasonal drought stress and vulnerability of overstory trees to extended drought in a mature Amazonian forest. Water Resources Research 48: W12507. doi:10.1029/2012WR011972
- Joetzjer E, Douville H, Delire C, Ciais P. 2013. Present-day and future Amazonian precipitation in global climate models: CMIP5 versus CMIP3. Climate Dynamics 41: 2921–2936. doi:10.1007/s00382-012-1644-1
- Kattge J, Díaz S, Lavorel S, Prentice IC, Leadley P, Bönisch G, Garnier E, Westoby M, Reich PB, Wright IJ, et al. 2011. TRY – a global database of plant traits. Global Change Biology 17: 2905–2935. doi:10.1111/j.1365-2486.2011.02451.x
- Kikuta SB, Richter H. 1992. Leaf discs or press saps? A Comparison of Techniques for the Determination of Osmotic Potentials in Freeze-Thawed Leaf Material. Journal of Experimental Botany 43: 1039–1044. doi:10.1093/jxb/43.8.1039
- Koide RT, Robichaux RH, Morse SR, Smith CM. 2000. Plant water status, hydraulic resistance and capacitance. In: Pearcy RW, Ehleringer JR, Mooney HA, Rundel PW, editors. Plant Physiological Ecology: Field Methods and Instrumentation. Netherlands: Springer. p. 161–183. Available from: http://link.springer.com/chapter/10.1007/978-94-009-2221-1_9. doi:10.1007/978-94-010-9013-1_9
- Kraft NJB, Valencia R, Ackerly DD. 2008. Functional traits and niche-based tree community assembly in an Amazonian forest. Science 322: 580–582. doi:10.1126/science.1160662
- Kursar TA, Engelbrecht BMJ, Burke A, Tyree MT, EI Omari B, Giraldo JP. 2009. Tolerance to low leaf water status of tropical tree seedlings is related to drought performance and distribution. Functional Ecology 23: 93–102. doi:10.1111/j.1365-2435.2008.01483.x
- Lasky JR, Bachelot B, Muscarella R, Schwartz N, Forero-Montaña J, Nytch CJ, Swenson NG, Thompson J, Zimmerman JK, Uriarte M. 2015. Ontogenetic shifts in trait-mediated mechanisms of plant community assembly. Ecology 96: 2157–2169. doi:10.1890/14-1809.1
- Lenz TI, Wright IJ, Westoby M. 2006. Interrelations among pressure–volume curve traits across species and water availability gradients. Physiologia Plantarum 127: 423–433. doi:10.1111/j.1399-3054.2006.00680.x
- Lewis SL, Brando PM, Phillips OL, Heijden GMF van der, Nepstad D. 2011. The 2010 Amazon drought. Science 331: 554–554. doi:10.1126/science.1200807
- Li L, McCormack ML, Ma C, Kong D, Zhang Q, Chen X, Zeng H, Niinemets Ü, Guo D. 2015. Leaf economics and hydraulic traits are decoupled in five species-rich tropical-subtropical forests. Ecology Letters 18: 899–906. doi:10.1111/ele.12466
- Liu J, Yunhong T, Slik JWF. 2014. Topography related habitat associations of tree species traits, composition and diversity in a Chinese tropical forest. Forest Ecology and Management 330: 75–81. doi:10.1016/j.foreco.2014.06.045
- Maréchaux I, Bartlett MK, Sack L, Baraloto C, Engel J, Joetzjer E, Chave J. 2015. Drought tolerance as predicted by leaf water potential at turgor loss point varies strongly across species within an Amazonian forest. Functional Ecology 29: 1268–1277. doi:10.1111/1365-2435.12452
- Marengo JA, Tomasella J, Alves LM, Soares WR, Rodriguez DA. 2011. The drought of 2010 in the context of historical droughts in the Amazon region. Geophysical Research Letters 38:L12703. doi:10.1029/2011GL047436
- Markesteijn L, Poorter L, Bongers F. 2007. Light-dependent leaf trait variation in 43 tropical dry forest tree species. American Journal of Botany 94: 515–525. doi:10.3732/ajb.94.4.515
- Markesteijn L, Poorter L, Bongers F, Paz H, Sack L. 2011a. Hydraulics and life history of tropical dry forest tree species: coordination of species’ drought and shade tolerance. New Phytologist 191: 480–495. doi:10.1111/j.1469-8137.2011.03708.x
- Markesteijn L, Poorter L, Paz H, Sack L, Bongers F. 2011b. Ecological differentiation in xylem cavitation resistance is associated with stem and leaf structural traits. Plant Cell and Environment 34: 137–148. doi:10.1111/j.1365-3040.2010.02231.x
- Markewitz D, Devine S, Davidson EA, Brando P, Nepstad DC. 2010. Soil moisture depletion under simulated drought in the Amazon: impacts on deep root uptake. New Phytologist 187: 592–607. doi:10.1111/j.1469-8137.2010.03391.x
- Marthews TR, Burslem DFRP, Paton SR, Yanyuez F, Mullins CE. 2008. Soil drying in a tropical forest: Three distinct environments controlled by gap size. Ecological Modelling 216: 369–384. doi:10.1016/j.ecolmodel.2008.05.011
- Martin-StPaul NK, Longepierre D, Huc R, Delzon S, Burlett R, Joffre R, Rambal S, Cochard H. 2014. How reliable are methods to assess xylem vulnerability to cavitation? The issue of “open vessel” artifact in oaks. Tree Physiology 34: 894–805. doi:10.1093/treephys/tpu059
- Meinzer FC, Andrade JL, Goldstein G, Holbrook NM, Cavelier J, Wright SJ. 1999. Partitioning of soil water among canopy trees in a seasonally dry tropical forest. Oecologia 121: 293–301. doi:10.1007/s004420050931
- Meinzer FC, Woodruff DR, Domec J-C, Goldstein G, Campanello PI, Gatti MG, Villalobos-Vega R. 2008. Coordination of leaf and stem water transport properties in tropical forest trees. Oecologia 156: 31–41. doi:10.1007/s00442-008-0974-5
- Nepstad DC, de Carvalho CR, Davidson EA, Jipp PH, Lefebvre PA, Negreiros GH, da Silva ED, Stone TA, Trumbore SE, Vieira S. 1994. The role of deep roots in the hydrological and carbon cycles of Amazonian forests and pastures. Nature 372: 666–669. doi:10.1038/372666a0
- Nepstad DC, Tohver IM, Ray D, Moutinho P, Cardinot G. 2007. Mortality of large trees and lianas following experimental drought in an Amazon forest. Ecology 88: 2259–2269. doi:10.1890/06-1046.1
- Niinemets Ü. 2010. Responses of forest trees to single and multiple environmental stresses from seedlings to mature plants: Past stress history, stress interactions, tolerance and acclimation. Forest Ecology and Management 260: 1623–1639. doi:10.1016/j.foreco.2010.07.054
- Niinemets Ü, Keenan TF, Hallik L. 2015. A worldwide analysis of within-canopy variations in leaf structural, chemical and physiological traits across plant functional types. New Phytologist 205: 973–993. doi:10.1111/nph.13096
- O’Brien MJ, Leuzinger S, Philipson CD, Tay J, Hector A. 2014. Drought survival of tropical tree seedlings enhanced by non-structural carbohydrate levels. Nature Climate Change 4: 710–714. doi:10.1038/nclimate2281
- Paine CET, Amissah L, Auge H, Baraloto C, Baruffol M, Bourland N, Bruelheide H, Daïnou K, Gouvenain RC, Doucet J-L, et al. 2015. Globally, functional traits are weak predictors of juvenile tree growth, and we do not know why. Journal of Ecology 103: 978–989. doi:10.1111/1365-2745.12401
- Paine CET, Baraloto C, Chave J, Hérault B. 2011. Functional traits of individual trees reveal ecological constraints on community assembly in tropical rain forests. Oikos 120: 720–727. doi:10.1111/j.1600-0706.2010.19110.x
- Pérez-Harguindeguy N, Díaz S, Garnier E, Lavorel S, Poorter H, Jaureguiberry P, Bret-Harte MS, Cornwell WK, Craine JM, Gurvich DE, et al. 2013. New handbook for standardised measurement of plant functional traits worldwide. Australian Journal of Botany 61: 167–234. doi:10.1071/BT12225
- Phillips OL, Aragao LEOC, Lewis LS, Fisher JB, Lloyd J, Lopez-Gonzalez G, Malhi Y, Monteagudo A, Peacock J, Quesada CA, et al. 2009. Drought sensitivity of the Amazon rainforest. Science 323: 1344–1347. doi:10.1126/science.1164033
- Phillips OL, van der Heijden G, Lewis SL, López-González G, Aragão LEOC, Lloyd J, Malhi Y, Monteagudo A, Almeida S, Dávila EA, et al. 2010. Drought-mortality relationships for tropical forests. New Phytologist 187: 631–646. doi:10.1111/j.1469-8137.2010.03359.x
- Poorter L, Bongers F, Sterck FJ, Wöll H. 2005. Beyond the regeneration phase: differentiation of height–light trajectories among tropical tree species. Journal of Ecology 93: 256–267. doi:10.1111/j.1365-2745.2004.00956.x
- R Core Team. 2013. R: A Language and Environment for Statistical Computing. Vienna, Austria: R Foundation for Statistical Computing. Available from: http://www.R-project.org/
- Reich PB. 2014. The world-wide “fast–slow” plant economics spectrum: a traits manifesto. Journal of Ecology 102: 275–301. doi:10.1111/1365-2745.12211
- Rowland L, da Costa ACL, Galbraith DR, Oliveira RS, Binks OJ, Oliveira A a. R, Pullen AM, Doughty CE, Metcalfe DB, Vasconcelos SS, et al. 2015. Death from drought in tropical forests is triggered by hydraulics not carbon starvation. Nature 528: 119–122. doi:10.1038/nature15539
- Rozendaal DMA, Hurtado VH, Poorter L. 2006. Plasticity in leaf traits of 38 tropical tree species in response to light; relationships with light demand and adult stature. Functional Ecology 20: 207–216. doi:10.1111/j.1365-2435.2006.01105.x
- Saatchi S, Asefi-Najafabady S, Malhi Y, Aragão LEOC, Anderson LO, Myneni RB, Nemani R. 2013. Persistent effects of a severe drought on Amazonian forest canopy. Proceedings of the National Academy of Sciences 110: 565–570. doi:10.1073/pnas.1204651110
- Sack L, Cowan PD, Jaikumar N, Holbrook NM. 2003. The “hydrology” of leaves: co-ordination of structure and function in temperate woody species. Plant Cell and Environment 26: 1343–1356. doi:10.1046/j.0016-8025.2003.01058.x
- Sack L, Holbrook NM. 2006. Leaf hydraulics. Annual Review of Plant Biology 57 361–381. doi:10.1146/annurev.arplant.56.032604.144141
- Sack L, Melcher PJ, Liu WH, Middleton E, Pardee T. 2006. How strong is intracanopy leaf plasticity in temperate deciduous trees? American Journal of Botany 93: 829–839. doi:10.3732/ajb.93.6.829
- Sack L, Pasquet-Kok J, PrometheusWiki contributors. 2011. Leaf pressure-volume curve parameters. In PrometheusWiki. Available from: http://www.publish.csiro.au/prometheuswiki/tiki-pagehistory.php?page=Leaf pressure-volume curve parameters&preview=16
- Sack L, Scoffoni C, John GP, Poorter H, Mason CM, Mendez-Alonzo R, Donovan LA. 2013. How do leaf veins influence the worldwide leaf economic spectrum? Review and synthesis. Journal of Experimental Botany 64: 4053–4080. doi:10.1093/jxb/ert316
- Sakschewski B, von Bloh W, Boit A, Rammig A, Kattge J, Poorter L, Peñuelas J, Thonicke K. 2015. Leaf and stem economics spectra drive diversity of functional plant traits in a dynamic global vegetation model. Global Change Biology 21: 2711–2725. doi:10.1111/gcb.12870
- Sala A, Hoch G. 2009. Height-related growth declines in ponderosa pine are not due to carbon limitation. Plant Cell and Environment 32: 22–30. doi:10.1111/j.1365-3040.2008.01896.x
- Sala A, Woodruff DR, Meinzer FC. 2012. Carbon dynamics in trees: feast or famine? Tree Physiology 32: 764–775. doi:10.1093/treephys/tpr143
- Scheiter S, Langan L, Higgins SI. 2013. Next-generation dynamic global vegetation models: learning from community ecology. New Phytologist 198: 957–969. doi:10.1111/nph.12210
- Scoffoni C, Rawls M, McKown A, Cochard H, Sack L. 2011. Decline of leaf hydraulic conductance with dehydration: relationship to leaf size and venation architecture. Plant Physiology 156: 832–843. doi:10.1104/pp.111.173856
- Siefert A, Violle C, Chalmandrier L, Albert CH, Taudiere A, Fajardo A, Aarssen LW, Baraloto C, Carlucci MB, Cianciaruso MV, et al. 2015. A global meta-analysis of the relative extent of intraspecific trait variation in plant communities. Ecology Letters 18: 1406-14019. doi:10.1111/ele.12508
- Silva CE, Kellner JR, Clark DB, Clark DA. 2013. Response of an old-growth tropical rainforest to transient high temperature and drought. Global Change Biology 19: 3423–3434. doi:10.1111/gcb.12312
- Stahl C, Burban B, Goret J-Y, Bonal D. 2011. Seasonal variations in stem CO2 efflux in the Neotropical rainforest of French Guiana. Annals of Forest Science 68: 771–782. doi:10.1007/s13595-011-0074-2
- Stahl C, Burban B, Wagner F, Goret J-Y, Bompy F, Bonal D. 2013. Influence of seasonal variations in soil water availability on gas exchange of tropical canopy trees. Biotropica 45: 155–164. doi:10.1111/j.1744-7429.2012.00902.x
- Stahl C, Herault B, Rossi V, Burban B, Brechet C, Bonal D. 2013. Depth of soil water uptake by tropical rainforest trees during dry periods: does tree dimension matter? Oecologia 173: 1191–1201. doi:10.1007/s00442-013-2724-6
- Tibshirani R, Leisch F. 2015. bootstrap: Functions for the Book “An Introduction to the Bootstrap”. R package version 2015.2. Available from: http://CRAN.R-project.org/package=bootstrap
- Torres-Ruiz JM, Cochard H, Mayr S, Beikircher B, Diaz-Espejo A, Rodriguez-Dominguez CM, Badel E, Fernández JE. 2014. Vulnerability to cavitation in Olea europaea current-year shoots: further evidence of an open-vessel artifact associated with centrifuge and air-injection techniques. Physiologia Plantarum 152: 465–474. doi:10.1111/ppl.12185
- Van Nieuwstadt MGL, Sheil D. 2005. Drought, fire and tree survival in a Borneo rain forest, East Kalimantan, Indonesia. Journal of Ecology 93: 191–201. doi:10.1111/j.1365-2745.2004.00954.x
- Violle C, Enquist BJ, McGill BJ, Jiang L, Albert CH, Hulshof C, Jung V, Messier J. 2012. The return of the variance: intraspecific variability in community ecology. Trends in Ecology & Evolution 27: 244–252. doi:10.1016/j.tree.2011.11.014
- Wagner F, Rossi V, Baraloto C, Bonal D, Stahl C, Hérault B. 2014. Are commonly measured functional traits involved in tropical tree responses to climate? International Journal of Ecology 2014: e389409. doi:10.1155/2014/389409
- Weerasinghe LK, Creek D, Crous KY, Xiang S, Liddell MJ, Turnbull MH, Atkin OK. 2014. Canopy position affects the relationships between leaf respiration and associated traits in a tropical rainforest in Far North Queensland. Tree Physiology 34: 564-584. doi:10.1093/treephys/tpu016
- Woodruff DR, Meinzer FC. 2011. Water stress, shoot growth and storage of non-structural carbohydrates along a tree height gradient in a tall conifer. Plant Cell and Environment 34: 1920–1930. doi:10.1111/j.1365-3040.2011.02388.x
- Wright SJ, Kitajima K, Kraft NJB, Reich PB, Wright IJ, Bunker DE, Condit R, Dalling JW, Davies SJ, Díaz S, et al. 2010. Functional traits and the growth–mortality trade-off in tropical trees. Ecology 91: 3664–3674. doi:10.1890/09-2335.1
- Wright SJ, Machado JL, Mulkey SS, Smith AP. 1992. Drought acclimation among tropical forest shrubs (Psychotria, Rubiaceae). Oecologia 89: 457–463. doi:10.1007/BF00317149
Attachments
Other
Dataset of estimated Pi_osm and Pi_tlp values of trees and saplings within tropical forest sites of French Guiana.
DownloadOther
Validation datapoints of the osmometer method for 6 Amazonian tree species.
Data were collected in Septembre 2015 within a tropical forest site in French Guiana (Nouragues, Petit Plateau).
Article statistics
 Views: 7108
Views: 7108
Downloads
 PDF: 875
PDF: 875
